Advertisements
Advertisements
Question
With a labelled diagram describe an activity to show the formation of an ester.
Solution
1 mL of ethanol and 1 mL of glacial acetic acid are taken in a test tube. 3-4 drops of concentrated sulphuric acid are then added to a test tube. The resulting mixture in a water bath is then heated (as shown in the figure) for 5 minutes. After this, the contents are poured in a beaker containing 20 mL water. A sweet-smelling compound (ethyl ethanoalte) will be obtained as the product.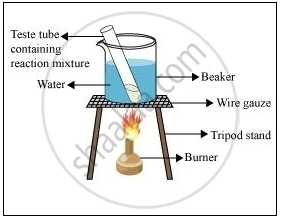
Formation of ester
When ethanoic acid reacts with ethanol in the presence of an acid, ethyl ethanoate is formed. Ethyl ethanoate is an ester and has a sweet smell.
APPEARS IN
RELATED QUESTIONS
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
Write a balanced chemical equations for Heating of Ethanol at 170°C in the presence of conc. Sulphuric acid
What is the common name of methanal?
What is meant by a functional group? Explain with an example.
Give the harmful effects of drinking alcohol.
When ethanol is heated with concentrated sulphuric acid at 170°C, it gets converted into ethene. In this reaction, concentrated sulphuric acid acts as:
(a) oxidising agent
(b) catalyst
(c) dehydrating agent
(d) reducing agent
Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
Choose the correct word/phrase from the options given below to complete the following sentence:
The product formed when ethene gas reacts with water in the presence of sulphuric acid is ______.
Write the equation for the following lab preparation:
Methanol from iodomethane.
Colorless ethanol is in liquid state at room temperature.
