Advertisements
Advertisements
Question
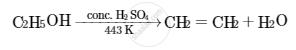
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
Solution
When ethanol (C2H5OH) is heated at 443 K with excess of conc. H2SO4, ethene (C2H4) is obtained. In this reaction, sulphuric acid acts as a dehydrating agent.
APPEARS IN
RELATED QUESTIONS
The compound formed when ethene reacts with Hydrogen is ________. (CH4, C2H6, C3H8)
What is the common name of methanal?
Give one exampleof the compound having the following functional group:
Alcohol group
What is meant by a functional group? Explain with an example.
Give the lab. Prepared of methyl alcohol.
How is the methylated spirit obtained?
Addition reactions and substitution reactions are types of organic reactions.Which type of reaction is shown by?
Ethene?
Convert ethanol into ethene using a solid dehydrating agent. Give only balanced equation.
Write the characteristics of ethanol.
How is ethene prepared from ethanol? Give the reaction involved in it.
