Advertisements
Advertisements
प्रश्न
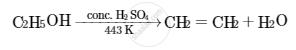
Write the name and structural formula of the compound obtained when ethanol is heated at 443 K with excess of conc. H2SO4. Also write chemical equation for the reaction stating the role of conc. H2SO4 in it.
उत्तर
When ethanol (C2H5OH) is heated at 443 K with excess of conc. H2SO4, ethene (C2H4) is obtained. In this reaction, sulphuric acid acts as a dehydrating agent.
APPEARS IN
संबंधित प्रश्न
How can ethanol and ethanoic acid be differentiated on the basis of their physical properties?
Write a balanced chemical equation for Burning of ethane in the plentiful supply of air.
What happens when (give chemical equation):
Sodium reacts with ethanol (ethyl alcohol)
What happens when ethanol reacts with ethanoic acid in the presence of a little of concentrated sulphuric acid? Write equation of the reaction involved.
What happens when ethanol is heated with concentrated sulphuric acid at 170°C? Write the equation of the reaction which takes place.
Give the structural formula of the following:
Ethanol
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above, name the homologue of the homologous series with the general formula CnH2n.
Convert ethanol into ethene using hot conc. H2SO4. Give only balanced equation.
State how the following conversion can be carried out:
Ethene to ethyl alcohol
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
