Advertisements
Advertisements
प्रश्न
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in colour
Give chemical equation for the reaction involved in the above case.
Give one example of a chemical reaction characterised by change in colour.
उत्तर
When sulphur dioxide gas is passed through acidified potassium dichromate solution, orange colour of potassium dichromate solution changes to green colour.
\[\ce{3SO2(g) + K2Cr2O7(aq) + H2SO4(aq)->K2SO4(aq) + Cr2(SO4)3(aq) + H2O(I)}\]
APPEARS IN
संबंधित प्रश्न
Give one example of an exothermic reaction.
Balance the following chemical equation:
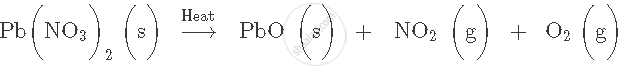
Balance the following equation. Also name the product formed.
` "H"_2"O"_2 → "H" _2 "O" + "O"_2`
Balance the following equation. Also name the product formed.
CO2 + C → CO
Balance the following equation:
Zn + KOH → K2ZnO2 + H2
Balance the following equation:
PbO + NH3 → Pb + H2O + N2
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – lead [IV] oxide.
What is meant by ‘reactants’ and ‘products’ in a chemical equation?
Give a balanced equation by the partial equation method, [steps are given below].
Oxidation of Lead [II] Sulphide by Ozone
- Ozone first decomposes to give molecular oxygen & nascent oxygen.
- Nascent oxygen then oxidises lead [II] sulphide to lead [II] sulphate.
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
3CuO + NH3 → 3Cu + 3H2O + N2
