Advertisements
Advertisements
प्रश्न
With the help of suitable examples explain the term neutralization and the formation of –
- acidic,
- basic and
- neutral salts
उत्तर
Neutralization is a type of chemical reaction in which an acid and base react to form salt and water with the evolution of heat.
- When the neutralization of a strong acid with a weak base takes place, it leads to the formation of acidic salt.
Example:
\[\ce{HCl +NH4OH -> NH4Cl +H2O}\] - When the neutralization of a weak acid with a strong base takes place, it leads to the formation of basic salt.
Example:
\[\ce{NaOH + CH3COOH -> CH3COO^-Na^+ + H2O}\] - When the neutralization of a strong acid with a strong base takes place, it leads to the formation of neutral salt.
Example:
\[\ce{NaOH + HCl -> NaCl + H2O}\]
APPEARS IN
संबंधित प्रश्न
When phenolphthalein is added to NaOH, the colour of the solution will become _________.
- colourless
- red
- pink
- yellow
Dry HCl gas does not change the colour of dry blue litmus paper. Why?
What happens when an acid reacts with a metal oxide? Explain with the help of an example. Write a balanced equation for the reaction involved.
What ions are present in the solutions of following substances? (write the symbols only)
Hydrochloric acid
What ions are present in the solutions of following substances? (write the symbols only)
Sulphuric acid
Explain why, sodium hydrogencarbonate is used as an antacid.
What is the chemical formula of bleaching powder?
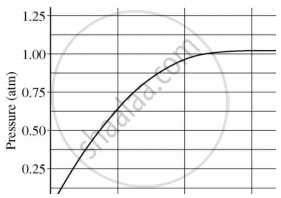
A student added 10 g of calcium carbonate in a rigid container, secured it tightly and started to heat it. After some time, an increase in pressure was observed, the pressure reading was then noted at intervals of 5 mins and plotted against time, in a graph as shown below. During which time interval did maximum decomposition take place?
What are strong and weak acids? In the following list of acids, separate strong acids from weak acids.
Hydrochloric acid, citric acid, acetic acid, nitric acid, formic acid, sulphuric acid.
Consider the following salt:
\[\ce{ZCO3}\]
What would be the change in colour in blue litmus if \[\ce{ZCO3}\] is added to it and Z is potassium?
