Advertisements
Advertisements
प्रश्न
Write chemical reactions taking place in the extraction of zinc from zinc blende.
उत्तर
The different steps involved in the extraction of zinc from zinc blende (ZnS) are given below:
(i) Concentration of ore
First, the gangue from zinc blende is removed by the froth floatation method.
(ii) Conversion to oxide (Roasting)
Sulphide ore is converted into oxide by the process of roasting. In this process, ZnS is heated in a regular supply of air in a furnace at a temperature, which is below the melting point of Zn.
`2 ZnS + 3O_2 -> 2ZnO + 2SO_2`
(iii) Extraction of zinc from zinc oxide (Reduction)
Zinc is extracted from zinc oxide by the process of reduction. The reduction of zinc oxide is carried out by mixing it with powdered coke and then, heating it at 673 K.
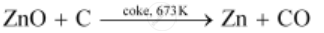
iv) Electrolytic Refining
Zinc can be refined by the process of electrolytic refining. In this process, impure zinc is made the anode while a pure copper strip is made the cathode. The electrolyte used is an acidified solution of zinc sulphate (ZnSO4). Electrolysis results in the transfer of zinc in pure from the anode to the cathode.
Anode : `Zn -> Zn^(2+) + 2e^(-)`
Cathode: `Zn^(2+) + 2e^(-) -> Zn`
APPEARS IN
संबंधित प्रश्न
How is 'cast iron' different from 'pig iron'?
The reaction,
Cr2O3 + 2Al → Al2O3 + 2Cr
(ΔGθ = -421kJ) is thermodynamically feasible as is apparent from the Gibbs energy value. Why does it not take place at room temperature?
Is it true that under certain conditions, Mg can reduce SiO2 and Si can reduce MgO? What are those conditions?
Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
How will you convert the following:
Zinc blende to Zinc metal
The impurity that is added externally to remove the impurity already present in the ore is known as ____________.
In the metallurgy of aluminium ______.
Choose the correct option of temperature at which carbon reduces \[\ce{FeO}\] to iron and produces \[\ce{CO}\].
Below point ‘A’ \[\ce{FeO}\] can ______.
For the metallurgical process of which of the ores calcined ore can be reduced by carbon?
(i) Haematite
(ii) Calamine
(iii) Iron pyrites
(iv) Sphalerite
Wrought iron is the purest form of iron. Write a reaction used for the preparation of wrought iron from cast iron. How can the impurities of sulphur, silicon and phosphorus be removed from cast iron?
How is copper extracted from low grade copper ores?
The mixture of compounds A and B is passed through a column of \[\ce{Al2O3}\] by using alcohol as eluant. Compound A is eluted in preference to compound B. Which of the compounds A or B, is more readily adsorbed on the column?
Explain the following:
Generally sulphide ores are converted into oxides before reduction.
Heating of MgCl2 6H2O in absence of HCl gives ______.
Carbon deposition reaction taking place in ______.
