Advertisements
Advertisements
प्रश्न
State the role of silica in the metallurgy of copper.
उत्तर १
During the roasting of pyrite ore, a mixture of FeO and Cu2O is obtained.
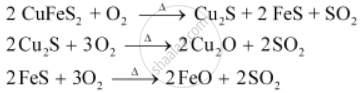
The role of silica in the metallurgy of copper is to remove the iron oxide obtained during the process of roasting as ‘slag’. If the sulphide ore of copper contains iron, then silica (SiO2) is added as flux before roasting. Then, FeO combines with silica to form iron silicate, FeSiO3 (slag).
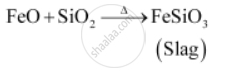
उत्तर २
During roasting, copper pyrites are converted into a mixture of FeO and Cu20. Thus, acidic flux silica is added during smelting to remove FeO (basic). FeO combines with SiO2to form famous silicate (FeSiO3) slag which floats over molten matte.
APPEARS IN
संबंधित प्रश्न
Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
Write chemical reactions taking place in the extraction of zinc from zinc blende.
The choice of a reducing agent in a particular case depends on thermodynamic factor. How far do you agree with this statement? Support your opinion with two examples.
In the extraction of copper from its sulphide ore, the metal is formed by the reduction of \[\ce{Cu2O}\] with ______.
Choose the correct option of temperature at which carbon reduces \[\ce{FeO}\] to iron and produces \[\ce{CO}\].
Below point ‘A’ \[\ce{FeO}\] can ______.
For the reduction of \[\ce{FeO}\] at the temperature corresponding to point D, which of the following statements is correct?
How is copper extracted from low grade copper ores?
The mixture of compounds A and B is passed through a column of \[\ce{Al2O3}\] by using alcohol as eluant. Compound A is eluted in preference to compound B. Which of the compounds A or B, is more readily adsorbed on the column?
Write down the reactions taking place in Blast furnace related to the metallurgy of iron in the temperature range 500-800 K.
Explain the following:
\[\ce{CO2}\] is a better reducing agent below 710 K whereas \[\ce{CO}\] is a better reducing agent above 710 K.
Explain the following:
Silica is added to the sulphide ore of copper in the reverberatory furnace.
Heating of MgCl2 6H2O in absence of HCl gives ______.
Carbon deposition reaction taking place in ______.
Assertion: For the extraction of iron, haematite ore is used.
Reason: Haematite is a carbonate ore of iron.
\[\ce{Au + CN^- + H2O + O2 -> [Au(CN)2]^- + OH^-}\]
The number of CN− ions involved in the balanced equation is ______.
