Advertisements
Advertisements
प्रश्न
Write the equations involved in the following reactions : Williamson synthesis
उत्तर १
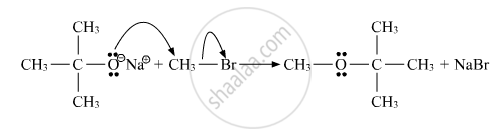
उत्तर २
Williamson synthesis: It involves the treatment of an alkyl halide with a suitable sodium alkoxide. The sodium alkoxide needed for the purpose is prepared by the action of sodium on a suitable alcohol. The reaction invlolves the nucleophillic displacement (substitution) of the halide ion from the alkyl halide by the alkoxide ion by SN2 mechanism.
R-OH +2Na → 2R’-O-Na+ + H2
R’-O-Na+ + R-X → R’-O-R +Na+ X
Ether
The second reaction is the substitution nucleophillic bimolecular (SN2) reaction.
For example:
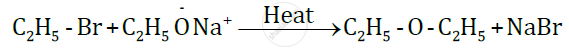
APPEARS IN
संबंधित प्रश्न
Explain the mechanism of the following reaction:
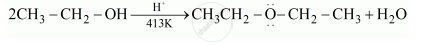
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Write the equations for the following reaction:
Tert butyl chloride is treated with sodium ethoxide.
Williamson's synthesis of ether is an example of ______.
Write the name of the reaction, structure and IUPAC name of the product formed when:
\[\ce{CH3CH2CH(CH3)CH(CH3) ONa}\] reacts with \[\ce{C2H5Br}\]
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
