Advertisements
Advertisements
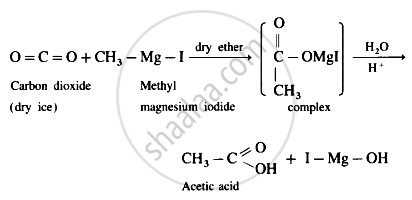
प्रश्न
Write preparation of acetic acid from:
dry ice
उत्तर
APPEARS IN
संबंधित प्रश्न
Answer in brief.
Write reaction showing the action of the following reagent on propanenitrile:
Dilute NaOH
What is the action of the following reagent on toluene?
Alkaline KMnO4, dil. HCl and heat
What is the action of the following reagent on toluene?
CrO2Cl2 in CS2
Write reaction showing conversion of p-bromoisopropylbenzene into p-isopropylbenzoic acid (3 steps).
Write reagent for conversion of alkyl nitrile into aldehyde.
Choose the CORRECT increasing order of the acidic strength of carboxylic acids from the following.
Identify X in the following reaction:

Identify the functional group in the product formed.
\[\begin{array}{cc}
\phantom{...........}\ce{O}\\
\phantom{...........}||\\
\ce{C2H5 - N - H + CH3 - C - Cl ->[Pyridine][-HCl]}\\
|\phantom{......................}\\
\ce{C2H5}\phantom{..................}
\end{array}\]
Hydrides of transition metals are used as powerful ____________ agents.
Acetic anhydride is obtained from acetyl chloride by the reaction of ____________.
Which is NOT present in Grignard reagent?
Identify the product C in following reaction.
\[\ce{CH3CH2COOH ->[NaOH] A ->[NaOH][CaO*\Delta] B ->[HNO3][\Delta] C}\]
Which of the following is obtained on alkaline hydrolysis of 1,1-dichlorocyclohexane?
Propane nitrile on reaction with ethyl magnesium iodide in presence of dry ether gives complex. This imine complex on acid hydrolysis forms.
Which of the following compounds on heating with potassium permanganate and dilute sulphuric acid forms adipic acid?
Identify product B in the following reaction.
\[\ce{C6H5MgBr ->[dry ice][dry ether] A ->[HOH][dil. HCl] B}\]
Write preparation of acetic acid from:
acetyl chloride
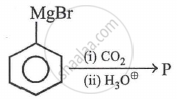
In the above reaction product 'P' is:
Which of the following alkenes on oxidation by KMnO4 in dil H2SO4 forms adipic acid?
Identify product B in the following reaction.
\[\ce{Cumene ->[KMnO4, KOH][\Delta] A ->[H3O+] B}\]
Write the reaction of Acetyl chloride on ethanamine.
