Advertisements
Advertisements
प्रश्न
Write the ionization of sulphuric acid showing the formation of hydronium ion.
उत्तर १
Ionization of sulphuric acid showing the formation of hydronium ion:
H2SO4 + H2O  H3O+ + HSO4-
H3O+ + HSO4-
HSO4- + H2O  H3O+ + SO42-
H3O+ + SO42-
उत्तर २
Water is a polar molecule which is represented as
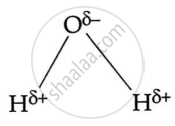
When acid dissolves in water, it interacts strongly with polar molecules, forming a proton [H+]. This proton cannot exist independently in an aqueous solution and attaches to a water molecule to create a hydrogen ion.
\[\ce{HCl <=>[H2O] H+ + Cl^1-}\]
\[\ce{H^1+ + H2O <=> \underset{(Hydronium ion)}{[H3O^1+]}}\]
or \[\begin{bmatrix}\begin{array}{cc}
\ce{H - \overset{\bullet\bullet}{\mathbf{O}} - H}\\
\downarrow\\
\ce{H}\\
\end{array}\end{bmatrix}^+\]
and \[\ce{H2SO4 + 2H2O <=> 2[H3O]+ + [SO4]^2-}\]
APPEARS IN
संबंधित प्रश्न
Identify the odd one out and justify.
Give a reason for the following.
Hydronium ions are always in the form H3O+.
Write the chemical equation for the following activity.
Dilute HCl was poured on baking soda.
| Source | Acid present | |
| 1. | Apple | ______ |
| 2. | ______ | Citric acid |
| 3. | ______ | tartaric acid |
| 4. | Tomato | ______ |
| 5. | Vinegar | ______ |
Explain the following.
H3PO3 is not a tribasic acid.
Explain the following:
H3PO3 is not a tribasic acid.
Explain the following:
\[\ce{H3PO3}\] is not a tribasic acid.
Explain the following:
H3PO3 is not a tribasic acid.
Explain the following:
\[\ce{H3PO3}\] is not a tribasic acid.
On which factor does the strength of an acid depend?
