Advertisements
Advertisements
प्रश्न
Write the names of isotopes of hydrogen.
उत्तर
Hydrogen has three naturally occuring isotopes namely Protium (1H1), Deuterium (1H2) and Tritium (1H3).
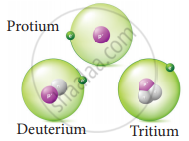
Isotopes of hydrogen
APPEARS IN
संबंधित प्रश्न
Explain the following.
Hydrogen shows similarity with alkali metals as well as halogens.
Answer the following question.
Outline the industrial production of dihydrogen from steam. Also, write the chemical reaction involved.
Name the following.
A compound having hydrogen, aluminium, and lithium as its constituent elements.
Choose the correct option.
Identify the odd one.
Which of the following statements about hydrogen is incorrect?
How do you convert para-hydrogen into ortho hydrogen?
An isotope of hydrogen (A) reacts with diatomic molecule of element which occupies group number 16 and period number 2 to give compound (B) is used as a moderator in nuclear reaction. (A) adds on to a compound (C), which has the molecular formula C3H6 to give (D). Identify A, B, C and D.
Which of the following statements is INCORRECT?
Caesium is used in ________.
The highest industrial consumption of molecular hydrogen is to produce compounds of the element:
