Advertisements
Advertisements
प्रश्न
Write three physical properties of acetic acid.
उत्तर
Three physical properties of acetic acid:
- State : Liquid
- Odour : Pleasant smell-smell of vinegar
- Taste : sour taste
APPEARS IN
संबंधित प्रश्न
Complete the following equation:
`CH_3 COOH + CH_2 H_5 OH` 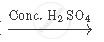
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
Explain the following term with an example.
Oxidant
Name the following:
A toxic alcohol
Fill in the blank with appropriate word/words.
Denatured alcohol is a mixture of _____ and _______
Draw the structural formula of a compound with two carbon atoms in the following case:
An alcohol containing two carbon atoms.
Using appropriate catalysts, ethane can be oxidized to an alcohol. Name the alcohol formed when ethane is oxidized.
Draw the structural diagram of:
Ethanoic acid
Observe the diagram given below and answer the questions:
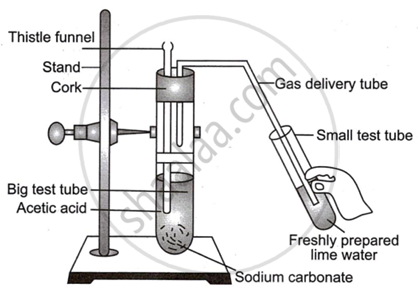
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
