Advertisements
Advertisements
प्रश्न
Write word equation for the following chemical reaction given below. Also state the observation seen in the case.
NaCl + AgNo3 → AgCU ↓ + NaN03
उत्तर
Sodium Chloride + Silver nitrate → Silver chloride + Sodium nitrate
AgCl (Silver Chloride) a white precipitate is formed in the above chemical reaction.
APPEARS IN
संबंधित प्रश्न
Balance the given equation:
HNO3 + Ca(OH)2  Ca(NO3)2 + H2O
Ca(NO3)2 + H2O
When hydrogen is passed over copper oxide, copper and steam are formed. Write a balanced equation for this reaction and state which of the chemicals are metals.
With the help of an appropriate example, justify that some of the chemical reactions are determined by Change in temperature.
Give chemical equation for the reaction involved in the above case.
Write your observation and name the product when ron nails are added to an aqueous solution of copper sulphate.
Write the balanced chemical equation of the following reaction. iron + sulphuric acid → ferrous sulphate + hydrogen.
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate moles of salt formed.
When a potassium iodide solution is added to a solution of lead (II) nitrate in a test tube, a precipitate is formed.
Write the balanced chemical equation for this reaction.
Balance the following simple equation:
H2 + O2 → H2O
Underline the compound in the equation given below, it is incorrectly balanced and write the correct balancing for the same.
CaC2 + N2 → 2CaCN2 + C
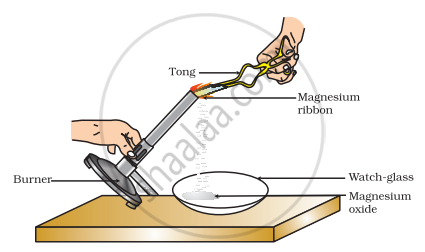
Which of the following is the correct observation of the reaction shown in the above set up?
