Advertisements
Advertisements
प्रश्न
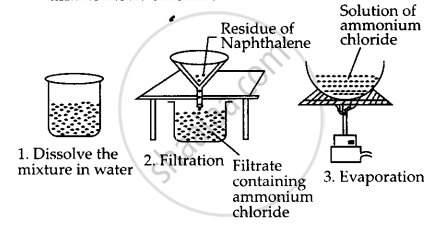
You are provided with a mixture of naphthalene and ammonium chloride by your teacher. Suggest an activity to separate them with well labelled diagram.
उत्तर
A mixture of naphthalene and ammonium chloride can be separated as follows :
Step-1: Put the mixture in a beaker and add water to it. Stir with a glass rod. Ammonium chloride is soluble in water gets dissolved leaving behind the insoluble naphthalene.
Step-2: Filter the solution. Naphthalene remains on the filter paper while ammonium chloride is obtained as filtrate.
Step-3: Evaporate the filtrate to get back ammonium chloride.
APPEARS IN
संबंधित प्रश्न
When sodium chloride is added to a definite volume of water and stirred well, a solution is formed, but there is no increase in the level of water. Why?
Write whether the following statement is true or false.
Only water can exist in three different states.
For the following statement, say whether it describe a solid, a liquid or a gas.
Particles are quite close together.
Fill in the blank:
The quantity of matter in an object is called its _____
Give an experiment to explain that there are intermolecular spaces between water molecules.
Write true or false for each statement. Rewrite the false statement correctly.
If the intermolecular space is more than the intermolecular force will be weaker.
Fill in the following blank with suitable words :
The smell of perfume gradually spreads across a room due to .....................
Explain the following :
When an incense stick is lighted in the corner of a room, its fragrance spreads quickly in the entire room.
Differentiate the general properties of solids, liquids and gases in the form of a table — with reference to shape.
Water obtained from tube wells is usually ______ water.
