Topics
Measurement
- Measurements
- Physical Quantities
- Unit and Its Types
- Unit Systems
- International System of Units (Si System)
- Base Quantities and Units
- Clocks
- Accuracy in Measurements
- Approximation
- Rounding off
Force and Pressure
- Force
- Effect of Force
- Thrust and Pressure
- Factors Affecting Pressure
- Atmospheric Pressure
- Measurement of Atmospheric Pressure
- Mercury Barometer (Simple Barometer)
- Buoyancy Force (Upthrust Force)
- Pressure of liquid
- Pascal’s Law
- Application of Pascal’s Law
- Surface Tension
- Viscous Force or Viscosity
- Force of Friction
- Kinds of Friction
- Advantages and Disadvantage of Friction
- Increasing and Reducing Friction
Light
- Light
- Mirrors
- Spherical Mirrors
- Images Formed by Spherical Mirrors
- Concave Mirror
- Convex Mirror
- Law of Reflection of Light
- Types of Reflection
- Multiple Reflections
- Introduction to Refraction of Light
- Refractive Index
- Prism
- Dispersion of Light Through Prism and Formation of Spectrum
Heat
- Heat and Its Unit
- Effects of Heat
- Transfer of Heat
- Conduction
- Convection
- Radiation
- Calorimetry and Calorimeter
- The Temperature and a Thermometer
- Scales of Thermometers
- Thermal Capacity (Heat Capacity)
- Specific Heat Capacity
- Thermostat
- Thermos Flask (Dewar Flask)
Electricity
- Electricity
- Structure of an Atom
- Electric Charge
- Electric Force
- Electric Field
- Transfer of Charges
- Direction of the Electric Current - Conventional and Electronic Flow
- Flow of Charges (Electrons) Between Conductor
- Electroscope
- Lightning and Lightning Safety
- Electric Circuit
- Types of Circuits: Simple Circuit
- Types of Circuits: Series Circuit
- Types of Circuits: Parallel Circuit
- Effects of Electric Current
- Heating Effect of Electric Current
- Chemical Effects of Electric Current
Sound
Magnetism
- Discovery of Magnets
- Magnet
- Classification of Magnets
- Magnetic Properties
- Magnetic Field
- Magnetic Materials
- Artificial Magnets
- Earth’s Magnetism
- Uses of Magnets
Universe and Space Science
- The Universe
- Galaxy and Its Types
- The Solar System
- Rockets and Its Structure
- Propellants and Its Types
- Space Launch Technology
- Indian Space Research Organization (ISRO)
- Concept of Space Missions
- India’s Space Programmes: Chandrayaan – 1
- India’s Space Programmes: Mangalyaan (Mars vehicle)
- India’s Space Programmes: Chandrayaan – 2
- Indian Scientist
- National Aeronautics and Space Administration (NASA)
- NASA Space Mission: Apollo Mission
- NASA’s work with ISRO
- Work of Indians at NASA
Matter Around Us
- Matter (Substance)
- Characteristics of Particles (Molecules) of Matter
- States of Matter
- The Solid State
- The Liquid State
- The Gaseous State
- Elements
- Symbols Used to Represent Atoms of Different Elements
- Types of Element: Metals
- Physical Properties of Metals
- Types of Element: Non-metal
- Physical Properties of Non-metal
- Type of Element: Metalloid
- Compound
- Types of Compound
Changes Around Us
- Chemical Reaction
- Factors Determining Chemical Changes
- Effects of Chemical Changes: Biological Effects
- Effects of Chemical Changes: Environmental Effects
- Effects of Chemical Changes: Production of Heat, Light, Sound and Pressure
Air
- Air Around Us
- Air is a Mixture
- Occurrence of Oxygen
- Properties of Oxygen
- Importance of Oxygen
- Occurrence of Nitrogen
- Properties of Nitrogen
- Importance of Nitrogen
- Nitrogen Fixation
- Composition and Components of Air
- Carbon Dioxide
- Properties of Carbon Dioxide
- Importance of Carbon Dioxide
- Green House Effect
- Preventive Measures of Green House Effect
- Global Warming
- Preventive Measures of Global Warming
- Acid Rain
- Preventive Measures of Acid Rain
Atomic Structure
- Atoms: Building Blocks of Matter
- Atoms: Building Blocks of Matter
- Dalton’s Atomic Theory
- Advantages and Limitations of Dalton’s Atomic Theory
- Discovery of Charged Particles in Matter
- Electrons (e)
- Protons (p)
- Neutrons (n)
- J. J. Thomson’s Atomic Model
- Advantage and Limitations of Thomson’s Atomic Model
- Valency
- Different Ways to Determine Valency
- Variable Valency
- Ions (Radicals) and Its Types
- Different Valent Ions
- Chemical Formula or Molecular Formula
- Naming Chemical Compounds
- Chemical Equation
- Balancing Chemical Equation
- Laws of Chemical Combination
- Law of Conservation of Mass
- Law of Constant Proportions (Law of Definite Proportions)
Water
- Water: Our Lifeline
- Composition of Water
- Electrolysis of Water
- Preparation of Water
- Physical Properties of Water
- Chemical Properties of Water
- Water - a Universal Solvent
- Potable Water
- Purification of Water
- Classification of water: Soft and Hard Water
- Advantage and Disadvantage of Hard Water
- Removal of Hardness of Water
- Water Pollution and Its Causes
- Prevention of Water Pollution
- Water Pollution in Tamilnadu
Acids and Bases
- Acids
- Properties of Acids
- Uses of Acids
- Bases (Alkalis)
- Properties of Bases (Alkalis)
- Uses of Bases
- Types of Double Displacement: Neutralization Reaction
- Neutralization Reactions in Our Daily Life
- Indicators
- Similarities and Differences Between Acids and Bases
Chemistry in Everyday Life
- Hydrocarbons
- Sources of Hydrocarbons
- Natural Gas
- Other Gases
- Non-crystalline/Amorphous Forms: Coal
- Extraction of Coal
- Non-crystalline/Amorphous Forms: Coke
- Petroleum
- Refining of Crude Petroleum
- Fuel
- Types of Fuel
- Fuel Efficiency
- Bio-energy
- Electricity Generation using Solar Energy
Microorganisms
- Microorganisms (Microbes) and Microbiology
- Microorganisms (Microbes) and Microbiology
- Viruses
- Bacteria
- Bacteria
- Fungi
- Algae
- Protozoa
- Prions
- Virions
- Useful micro-organisms
- Harmful Microorganisms
- Relationship Between Man and Microbes Balances, Imbalances and Uses
Plant Kingdom
- Biodiversity
- Kingdom Plantae: Thallophyta (Algae)
- Kingdom Plantae: Thallophyta (Fungi)
- Division II- Bryophytes
- Division III- Pteridophytes
- Division I-Gymnosperms
- Division II- Angiosperms
- Taxonomy and Systematics
- Nomenclature
- Medicinal Plant
Organisation of Life
- Biological Organisation in Organisms
- Cell: Structural and Functional Unit of Life
- Organisms Show Variety in Cell Number, Shape and Size
- Tissues - “The Teams of Workers”
- Human Organ System
- Human Eye
- Human Respiratory System
- Mechanism of respiration-Breathing
- Physiological Processes in Organisms
- Homeostasis
- Simple Diffusion
- Concept of Osmosis
- Osmotic Pressure
- Osmoregulation
- Types of Respiration: Aerobic and Anaerobic Respiration
- Metabolism
Movements in Animals
- Biodiversity
- Movement and Locomotion
- Movement in Different Animals
- Human Body and Its Movements
- Types of Movements
- Joints and Its Classification
- Human Skeleton System
- The Human Skeleton: Axial Skeleton
- The Human Skeleton: Appendicular Skeleton
- Muscles and Its Types
- Muscular System
Reaching the Age of Adolescence
- Adolescence and Puberty
- Changes at Puberty
- Secondary Sex Characteristics
- Role of Hormones in Initiating Reproductive Function
- Reproductive Phase of Life in Humans
- Menstrual Cycle (Ovarian Cycle)
- Reproductive Health
- Nutritional Needs of Adolescents
- Personal Hygiene for Adolescence
Crop Production and Management
- Crop and Its Types
- The Green Revolution
- Soil Formation and Preparation for Agriculture
- Agricultural Implements
- Fertilizers
- Food Storage and Conservation of the Environment
- Manuring (Biomanuring)
- Fertilizers
- Methods to Replenish Nutrients in Your Soil
- Improved methods of agriculture
- Food Security
- Agricultural Assistance Programme
- Weeding
- Harvesting of Crops
- Storage of Food Grains
- Rotation of Crops
- Seed Bank
- Bio-indicators
- Agriculture Research Institutions
- Foliar Sprays
- Bio-control Methods
Conservation of Plants and Animals
- Deforestation and Its Causes
- Consequences of Deforestation
- Afforestation
- Reforestation
- Classification of Threatened Species
- Causes for Endangerment
- Saving Endangered Species
- Red Data Book
- Conservation of Forest
- Conservation of Wildlife
- PBR (People’s Bio-diversity Register)
- Biomagnification
- Animal Welfare Organisations
Libre Office Calc
- Libre Office Calc
- Spreadsheet
- Worksheet and Workbook
- Merge Cells
- Generate Auto Numbers
- Functions
- Operators in Libre Office Calc
- Working with Charts
- Characteristics of particles or matter
- Classification of Matter and Particle
- Experiment
Characteristics of particles or matter:
1. Particles of matter have space between them
Particles of one type of matter get into the spaces between particles of the other. This shows that there is enough space between particles of matter.

2. Particles of matter are continuously moving
Particles of matter are continuously moving as they have kinetic energy. With the increase in temperature, the kinetic energy of the particles also increases and particles move faster.
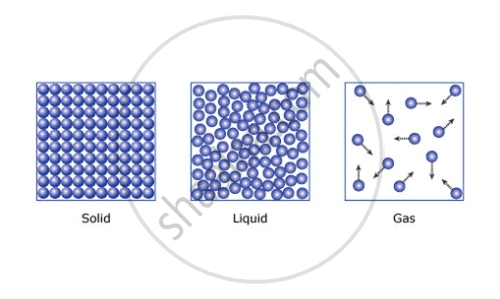
3. Particles of matter attract each other
Particles of matter have a force acting between them. This force keeps the particles together. The strength of this force of attraction varies from one kind of matter to another
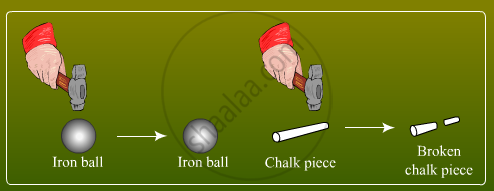
From the above figure, it is obvious that it would be easier to break a piece of chalk than the iron ball. This is because the particles in the iron ball are held together with greater force than that of the chalk.
Classification of Matter and Particle:
- Solids: Particles are very close together, vibrating in fixed positions. This gives solids a definite shape and volume, high density, and makes them non-compressible.
- Liquids: Particles are still close but can move past each other. The intermolecular force is moderate, allowing liquids to have a definite volume but no fixed shape, taking the shape of their container. Liquids also have fluidity.
- Gases: Particles are far apart with very weak intermolecular forces, moving freely and occupying all available space. Gases have no definite shape or volume.
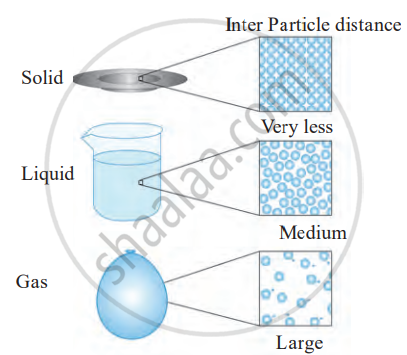
Physical state of matter: submicroscopic picture
| Physical state of matter | Fluidity / Rigidity / Plasticity / Elasticity | Volume | Shape | Compressibility | Intermolecular force | Distance between particles |
|---|---|---|---|---|---|---|
| Solid | Rigid/plastic/elastic | Definite | Definite | Negligible | Strong | Minimum |
| Liquid | Fluid | Definite | Indefinite | Very small | Moderate | Moderate |
| Gaseous | Fluid | Indefinite | Indefinite | Very high | Very weak | Very large |
Classification by Composition
Matter can be classified into elements, compounds, and mixtures based on the type of smallest particles:
- Element: Made of identical atoms (e.g., an oxygen molecule, O₂, has two identical oxygen atoms).
- Compound: Made of different atoms bonded together (e.g., a water molecule, H₂O, has two hydrogen atoms and one oxygen atom).
- Mixture: Contains a combination of different atoms or molecules (e.g., air has N₂, O₂, CO₂, etc.; brass is a mix of copper and zinc atoms).
Experiment
1. Aim: To observe how energy affects the movement of particles, similar to how particles behave in solid, liquid, and gaseous states.
2. Requirements: mustard seeds, a transparent plastic jar, a large balloon, a long thread, a needle, and a rubber band.
3. Procedure:
- Take some mustard seeds and place them in a transparent plastic jar.
- Thread a long thread through the centre of a large balloon using a needle and tie it securely.
- Stretch the balloon over the mouth of the jar, creating a rubber diaphragm, and fix it with a rubber band.
| Mode of pulling the diaphragm up and down | Movement of the mustard seeds |
|---|---|
| Slow | Vibration in the same place |
| With moderate force | Slight movement from their position |
| Vigorous | Rapid and random movement throughout |
 Movement of mustard seeds
Movement of mustard seeds
4. Conclusion: The movement of mustard seeds changes depending on how much energy is applied. This demonstrates how particles in solids, liquids, and gases move differently based on the energy they receive and the intermolecular forces acting between them.
