Topics
Physical World and Measurement
Physical World
Units and Measurements
- International System of Units
- Measurement of Length
- Measurement of Mass
- Measurement of Time
- Accuracy, Precision and Least Count of Measuring Instruments
- Errors in Measurements
- Significant Figures
- Dimensions of Physical Quantities
- Dimensional Formulae and Dimensional Equations
- Dimensional Analysis and Its Applications
- Need for Measurement
- Units of Measurement
- Fundamental and Derived Units
- Length, Mass and Time Measurements
- Introduction of Units and Measurements
Motion in a Straight Line
- Position, Path Length and Displacement
- Average Velocity and Average Speed
- Instantaneous Velocity and Speed
- Kinematic Equations for Uniformly Accelerated Motion
- Acceleration (Average and Instantaneous)
- Relative Velocity
- Elementary Concept of Differentiation and Integration for Describing Motion
- Uniform and Non-uniform Motion
- Uniformly Accelerated Motion
- Position-time, Velocity-time and Acceleration-time Graphs
- Position - Time Graph
- Relations for Uniformly Accelerated Motion (Graphical Treatment)
- Introduction of Motion in One Dimension
- Motion in a Straight Line
Kinematics
Motion in a Plane
- Scalars and Vectors
- Multiplication of Vectors by a Real Number or Scalar
- Addition and Subtraction of Vectors - Graphical Method
- Resolution of Vectors
- Vector Addition – Analytical Method
- Motion in a Plane
- Motion in a Plane with Constant Acceleration
- Projectile Motion
- Uniform Circular Motion (UCM)
- General Vectors and Their Notations
- Motion in a Plane - Average Velocity and Instantaneous Velocity
- Rectangular Components
- Scalar (Dot) and Vector (Cross) Product of Vectors
- Relative Velocity in Two Dimensions
- Cases of Uniform Velocity
- Cases of Uniform Acceleration Projectile Motion
- Motion in a Plane - Average Acceleration and Instantaneous Acceleration
- Angular Velocity
- Introduction of Motion in One Dimension
Laws of Motion
Work, Energy and Power
Laws of Motion
- Aristotle’s Fallacy
- The Law of Inertia
- Newton's First Law of Motion
- Newton’s Second Law of Motion
- Newton's Third Law of Motion
- Conservation of Momentum
- Equilibrium of a Particle
- Common Forces in Mechanics
- Circular Motion and Its Characteristics
- Solving Problems in Mechanics
- Static and Kinetic Friction
- Laws of Friction
- Inertia
- Intuitive Concept of Force
- Dynamics of Uniform Circular Motion - Centripetal Force
- Examples of Circular Motion (Vehicle on a Level Circular Road, Vehicle on a Banked Road)
- Lubrication - (Laws of Motion)
- Law of Conservation of Linear Momentum and Its Applications
- Rolling Friction
- Introduction of Motion in One Dimension
Work, Energy and Power
- Introduction of Work, Energy and Power
- Notions of Work and Kinetic Energy: the Work-energy Theorem
- Kinetic Energy (K)
- Work Done by a Constant Force and a Variable Force
- Concept of Work
- Potential Energy (U)
- Conservation of Mechanical Energy
- Potential Energy of a Spring
- Various Forms of Energy : the Law of Conservation of Energy
- Power
- Collisions
- Non - Conservative Forces - Motion in a Vertical Circle
Motion of System of Particles and Rigid Body
System of Particles and Rotational Motion
- Motion - Rigid Body
- Centre of Mass
- Motion of Centre of Mass
- Linear Momentum of a System of Particles
- Vector Product of Two Vectors
- Angular Velocity and Its Relation with Linear Velocity
- Torque and Angular Momentum
- Equilibrium of Rigid Body
- Moment of Inertia
- Theorems of Perpendicular and Parallel Axes
- Kinematics of Rotational Motion About a Fixed Axis
- Dynamics of Rotational Motion About a Fixed Axis
- Angular Momentum in Case of Rotation About a Fixed Axis
- Rolling Motion
- Momentum Conservation and Centre of Mass Motion
- Centre of Mass of a Rigid Body
- Centre of Mass of a Uniform Rod
- Rigid Body Rotation
- Equations of Rotational Motion
- Comparison of Linear and Rotational Motions
- Values of Moments of Inertia for Simple Geometrical Objects (No Derivation)
Gravitation
Gravitation
- Kepler’s Laws
- Newton’s Universal Law of Gravitation
- The Gravitational Constant
- Acceleration Due to Gravity of the Earth
- Acceleration Due to Gravity Below and Above the Earth's Surface
- Acceleration Due to Gravity and Its Variation with Altitude and Depth
- Gravitational Potential Energy
- Escape Speed
- Earth Satellites
- Energy of an Orbiting Satellite
- Geostationary and Polar Satellites
- Weightlessness
- Escape Velocity
- Orbital Velocity of a Satellite
Properties of Bulk Matter
Mechanical Properties of Solids
- Elastic Behaviour of Solid
- Stress and Strain
- Hooke’s Law
- Stress-strain Curve
- Young’s Modulus
- Determination of Young’s Modulus of the Material of a Wire
- Shear Modulus or Modulus of Rigidity
- Bulk Modulus
- Application of Elastic Behaviour of Materials
- Elastic Energy
- Poisson’s Ratio
Thermodynamics
Behaviour of Perfect Gases and Kinetic Theory of Gases
Mechanical Properties of Fluids
- Thrust and Pressure
- Pascal’s Law
- Variation of Pressure with Depth
- Atmospheric Pressure and Gauge Pressure
- Hydraulic Machines
- Streamline and Turbulent Flow
- Applications of Bernoulli’s Equation
- Viscous Force or Viscosity
- Reynold's Number
- Surface Tension
- Effect of Gravity on Fluid Pressure
- Terminal Velocity
- Critical Velocity
- Excess of Pressure Across a Curved Surface
- Introduction of Mechanical Properties of Fluids
- Archimedes' Principle
- Stoke's Law
- Equation of Continuity
- Torricelli's Law
Oscillations and Waves
Thermal Properties of Matter
- Heat and Temperature
- Measurement of Temperature
- Ideal-gas Equation and Absolute Temperature
- Thermal Expansion
- Specific Heat Capacity
- Calorimetry
- Change of State - Latent Heat Capacity
- Conduction
- Convection
- Radiation
- Newton’s Law of Cooling
- Qualitative Ideas of Black Body Radiation
- Wien's Displacement Law
- Stefan's Law
- Anomalous Expansion of Water
- Liquids and Gases
- Thermal Expansion of Solids
- Green House Effect
Thermodynamics
- Thermal Equilibrium
- Zeroth Law of Thermodynamics
- Heat, Internal Energy and Work
- First Law of Thermodynamics
- Specific Heat Capacity
- Thermodynamic State Variables and Equation of State
- Thermodynamic Process
- Heat Engine
- Refrigerators and Heat Pumps
- Second Law of Thermodynamics
- Reversible and Irreversible Processes
- Carnot Engine
Kinetic Theory
- Molecular Nature of Matter
- Gases and Its Characteristics
- Equation of State of a Perfect Gas
- Work Done in Compressing a Gas
- Introduction of Kinetic Theory of an Ideal Gas
- Interpretation of Temperature in Kinetic Theory
- Law of Equipartition of Energy
- Specific Heat Capacities - Gases
- Mean Free Path
- Kinetic Theory of Gases - Concept of Pressure
- Assumptions of Kinetic Theory of Gases
- RMS Speed of Gas Molecules
- Degrees of Freedom
- Avogadro's Number
Oscillations
- Periodic and Oscillatory Motion
- Simple Harmonic Motion (S.H.M.)
- Simple Harmonic Motion and Uniform Circular Motion
- Velocity and Acceleration in Simple Harmonic Motion
- Force Law for Simple Harmonic Motion
- Energy in Simple Harmonic Motion
- Some Systems Executing Simple Harmonic Motion
- Damped Simple Harmonic Motion
- Forced Oscillations and Resonance
- Displacement as a Function of Time
- Periodic Functions
- Oscillations - Frequency
- Simple Pendulum
Waves
- Reflection of Transverse and Longitudinal Waves
- Displacement Relation for a Progressive Wave
- The Speed of a Travelling Wave
- Principle of Superposition of Waves
- Introduction of Reflection of Waves
- Standing Waves and Normal Modes
- Beats
- Doppler Effect
- Wave Motion
- Speed of Wave Motion
- Surface Tension
- Force due to surface tension
- Factors affecting surface tension
1) Nature of liquid
2) Impurities
3) Temperature
4) Electrification - Applications of surface tension
Notes
Surface Energy
-
Surface energy is the excess energy exhibited by the liquid molecules on the surface compared to those inside the liquid.
-
This means liquid molecules at the surface have greater energy as compared to molecules inside it.
-
Suppose there is a tumbler and when we pour water in the tumbler, it takes the shape of the tumbler.
-
It acquires free surface.
Case 1: When molecules are inside the liquid:-
-
Suppose there is a molecule inside the water, there will be several other molecules that will attract that molecule in all the directions.
-
As a result this attraction will bind all the molecules together.
-
This results in negative potential energy of the molecule as it binds the molecule.
-
To separate this molecule huge amount of energy is required to overcome potential energy.
-
Some external energy is required to move this molecule and it should be greater than the potential energy.
-
Therefore in order to separate this molecule a huge amount of energy is required.
Case2: When the molecules are at the surface:-
- When the molecule is at the surface, half of it will be inside and half of it is exposed to the atmosphere.
-
For the lower half of the molecule it will be attracted by the other molecules inside the liquid.
-
But the upper half is free. The negative potential energy is only because of lower half.
-
But the magnitude is half as compared to the potential energy of the molecule which is fully inside the liquid.
-
So the molecule has some excess energy, because of this additional energy which the molecules have at the surface different phenomenon happen like surface energy, surface tension.
-
Liquids always tend to have least surface are when left to itself.
-
As more surface area will require more energy as a result liquids tend to have least surface area.
Surface energy for two fluids in contact:
-
Whenever there are two fluids, in contact, surface energy depends on materials of the surfaces in contact.
-
Surface energy decreases if the molecules of the two fluids attract.
-
Surface energy increases if molecules of the two fluids repel.
Surface Tension
-
Surface tension is the property of the liquid surface which arises due to the fact that surface molecules have extra energy.
-
Surface energy is the extra energy that the molecules at the surface have.
-
Surface tension is the property of the liquid surface because the molecules have extra energy.
-
Surface energy is defined as surface energy per unit area of the liquid surface.
-
Denoted by ’S’.
Mathematically:-
-
Consider a case in which liquid is enclosed in a movable bar.
-
Slide the bar slightly and it moves some distance (‘d’).
-
There will be increase in the area, (dl) where l=length of the bar.
-
Liquids have two surfaces one on the bar and others above the bar. Therefore area=2(dl)
-
Work done for this change =F x displacement.
-
Surface tension(S)=
-
Or Surface Energy=S x area
-
=S x 2dl
-
Therefore S x 2dl =F x d
-
-
Surface tension is the surface energy per unit area of the liquid surface.
-
It can be also defined as Force per unit length on the liquid surface.
-
Important: -At any interface (it is a line that separates two different medium) the surface tension always acts in the equal and opposite direction and it is always perpendicular to the line at the interface.
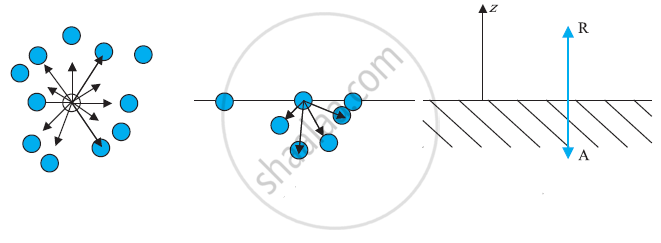 a) A molecule inside a liquid. Forces on a molecule due to others are shown. The direction of arrows indicates the attraction of repulsion. (b) Same, for a molecule at a surface. (c) Balance of attractive (A) and repulsive (R) forces.
a) A molecule inside a liquid. Forces on a molecule due to others are shown. The direction of arrows indicates the attraction of repulsion. (b) Same, for a molecule at a surface. (c) Balance of attractive (A) and repulsive (R) forces.
Surface tension and Surface energy: practical applications
-
Consider a molecule that is present completely inside the liquid and if it is strongly attracted by the neighboring molecules then the surface energy is less.
-
Consider a molecule that is present partially inside the liquid the force of attraction by the neighboring molecules is lesser as a result surface energy is more.
-
Consider a molecule whose very little part is inside the water so very small force of attraction by the neighboring molecules as a result of more surface energy.
-
Conclusion: - A fluid will stick to a solid surface if the surface energy between fluid and solid is smaller than the sum of energies between solid-air and fluid-air.
This means Ssf( solid-fluid) < Sfa(fluid air) + Ssa(Solid air)
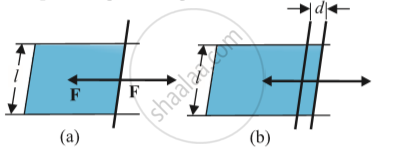
Stretching a film:
(a) A film in equilibrium
(b) The film stretched an extra distance
Why does water stick to glass but Mercury doesn’t?
In case of water and glass, water sticks to glass because the surface energy of water and glass is less than the surface energy between water and air and between glass and air.
In case of mercury, surface energy between mercury and glass
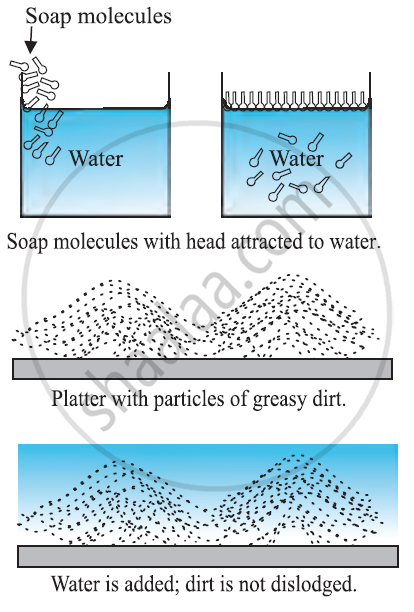
How detergents work?
-
Washing alone with the water can remove some of the dirt but it does not remove the grease stains. This is because water does not wet greasy dirt.
-
We need detergent which mixes water with dirt to remove it from the clothes.
-
Detergent molecules look like hairpin shape. When we add detergents to the water one end stick to water and the other end sticks to the dirt.
-
As a result dirt is getting attracted to the detergent molecules and they get detached from the clothes and they are suspended in the water.
-
Detergent molecules get attracted to water and when water is removed the dirt also gets removed from the clothes.

Angle of Contact:-
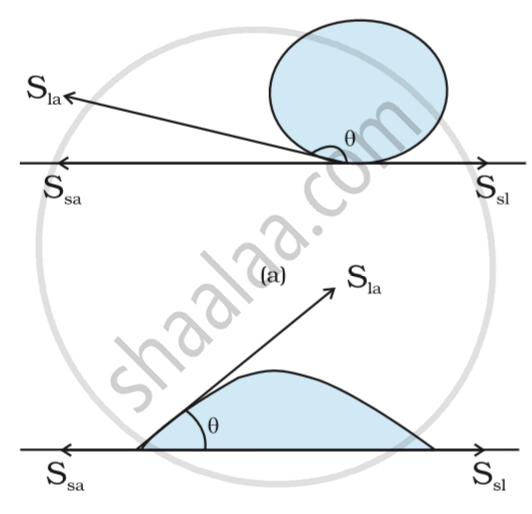
- The angle of contact is the angle at which a liquid interface meets a solid surface.
-
It is denoted by θ.
-
It is different at interfaces of different pairs of liquids and solids.
-
For example - Droplet of water on a lotus leaf. The droplet of water(Liquid) is in contact with the solid surface which is a leaf.
-
This liquid surface makes some angle with the solid surface. This angle is known as angle of contact.

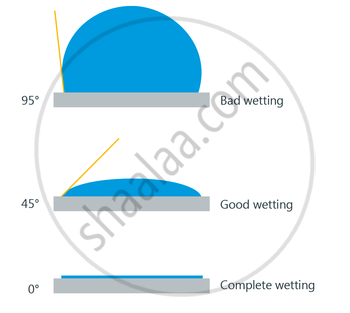
Significance of Angle of Contact
-
Angle of contact determines whether a liquid will spread on the surface of a solid or it will form droplets on it.
-
If the Angle of contact is obtuse: then droplet will be formed.
-
If the Angle of contact is acute: then the water will spread.
-
Case1: When droplet is formed
-
Consider we have a solid surface, droplet of water which is liquid and air.
-
The solid liquid interface denoted by Ssl, solid air interface denoted by Ssa and liquid air interface denoted by Sla.
-
The angle which Ssl makes with Sla. It is greater than the 900.
-
Therefore droplet is formed.
Case 2: When water just spreads
-
The angle which liquid forms with solid surface is less than 900.
Drops and Bubbles
Why water and bubbles are drops?
-
Whenever liquid is left to itself it tends to acquire the least possible surface area so that it has the least surface energy so it has the most stability.
-
Therefore for more stability, they acquire the shape of the sphere, as a sphere has the least possible area.
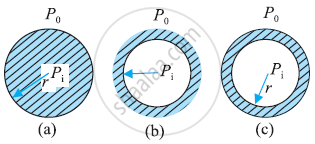
Distinction between Drop, Cavity, and Bubble:
Drop: - Drop is a spherical structure filled with water:
There is only one interface in the drop.
The interface separates water and air.
Example: Water droplet.
Water droplets:
Cavity:- Cavity is a spherical shape filled with air.
In the surroundings there is water and in the middle, there is a cavity filled with air.
There is only one interface which separates air and water.
Example:- bubble inside the aquarium.
Bubble:- In a bubble there are two interfaces. One is air-water and another is water and air.
Inside a bubble there is air and there is air outside.
But it consists of a thin film of water.
Pressure inside a drop and a cavity
-
The pressure inside a drop is greater than the pressure outside.
-
Suppose there is a spherical drop of water of radius ‘r’ which is in equilibrium.
-
Consider there is an increase in radius which is Δr.
Therefore extra surface energy = surface tension(S) x area
after calculating
extra surface energy =
At equilibrium, extra surface energy = energy gain due to the pressure difference
where
After calculation
Pressure inside a Bubble
-
Pressure inside a bubble is greater than the pressure outside.
- As bubble has 2 interfaces,
- This is probably why you have to blow hard, but not too hard, to form a soap bubble. A little extra air pressure is needed inside!
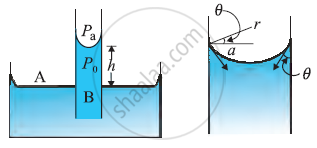
Capillary Rise
-
In Latin the word capilla means hair.
-
Due to the pressure difference across a curved liquid-air interface the water rises up in a narrow tube in spite of gravity.
-
Consider a vertical capillary tube of circular cross section (radius a) inserted into an open vessel of water.
-
The contact angle between water and glass is acute. Thus the surface of water in the capillary is concave. As a result there is a pressure difference between the two sides of the top surface. This is given by
Thus the pressure of the water inside the tube, just at the meniscus is less than the atmospheric pressure.
consider the two points A and B. They must be at the same pressure.
Where
By using eq (i) & (ii)
Therefore the capillary rise is due to surface tension. It is larger, for a smaller radius.
Problem:- The lower end of a capillary tube of diameter 2.00 mm is dipped 8.00 cm below the surface of the water in a beaker. What is the pressure required in the tube in order to blow a hemispherical bubble at its end in the water? The surface tension of water at temperature of the experiments is 7.30 ×10-2 Nm-1. 1 atmospheric pressure = 1.01 × 105 Pa, density of water = 1000 kg/m3, g = 9.80 m s-2.Also, calculate the excess pressure.
Answer:-
The excess pressure in a bubble of gas in a liquid is given by
Therefore, the pressure inside the bubble is
