Topics
Physics and Measurement
- What is Physics?
- Scope and Excitement of Physics
- Physics Related to Technology and Society
- Fundamental Forces in Nature
- Nature of Physical Laws
- Physical Quantities
- Unit and Its Types
- Unit Systems
- International System of Units (Si System)
- Unit Prefixes
- Measurement of Length
- Measurement of Mass
- Measurement of Time
- Accuracy, Precision and Least Count of Measuring Instruments
- Errors in Measurements
- Significant Figures
- Dimensions of Physical Quantities
- Dimensional Formulae and Dimensional Equations
- Dimensional Analysis and Its Applications
Kinematics
- Introduction to Kinematics
- Position, Path Length and Displacement
- Position - Time Graph
- Speed and Velocity
- Uniform and Non-uniform Motion
- Average Speed and Instantaneous Velocity
- Uniformly Accelerated Motion
- Velocity - Time Graphs
- Relations for Uniformly Accelerated Motion (Graphical Treatment)
- Scalars and Vectors
- Types of Vectors
- Addition and Subtraction of Vectors - Graphical Method
- Vector Addition – Analytical Method
- Scalar (Dot) and Vector (Cross) Product of Vectors
- Resolution of Vectors
- Relative Velocity
- Motion in a Plane
- Projectile Motion
- Uniform Circular Motion (UCM)
- Motion Under Gravity
- Motion in a Plane with Constant Acceleration
- Relative Velocity in Two Dimensions
Laws of Motion
- Introduction to Laws of Motion
- Aristotle’s Fallacy
- The Law of Inertia
- Newton's First Law of Motion
- Newton's Second Law of Motion
- Newton's Third Law of Motion
- Conservation of Momentum
- Law of Conservation of Linear Momentum and Its Applications
- Equilibrium of a Particle
- Common Forces in Mechanics
- Rolling Friction
- Circular Motion and Its Characteristics
- Static and Kinetic Friction
- Laws of Friction
- Dynamics of Uniform Circular Motion - Centripetal Force
- Solving Problems in Mechanics
- Motion of Connected Bodies, Pulley and Equilibrium of Forces
- Friction
- Banking of Roads
Work, Energy, and Power
- Introduction of Work, Energy and Power
- Notions of Work and Kinetic Energy: the Work-energy Theorem
- Concept of Work
- Kinetic Energy (K)
- Concept of Work
- Work Done by a Constant Force and a Variable Force
- The Work-energy Theorem for a Variable Force
- Potential Energy (U)
- Conservation of Mechanical Energy
- Potential Energy of a Spring
- Various Forms of Energy : the Law of Conservation of Energy
- Power
- Collisions
- The Concept of Energy
Rotational Motion
- Centre of Mass of Two-particle System
- Centre of Mass of a Rigid Body
- Basic Concepts of Rotational Motion
- Moment of a Force
- Torque and Angular Momentum
- Moment of Inertia
- Values of Moments of Inertia for Simple Geometrical Objects (No Derivation)
- Theorems of Perpendicular and Parallel Axes
- Rigid Body Rotation
- Equations of Rotational Motion
- Centre of Gravity
- Principle of Moments
- Angular Displacement
- Velocity and Acceleration in Simple Harmonic Motion
- Couple and Its Torque
- Rolling Motion
- Rotational K.E.
Gravitation
- Introduction to Gravitation
- Kepler’s Laws
- Newton’s Universal Law of Gravitation
- The Gravitational Constant
- Acceleration Due to Gravity of the Earth
- Acceleration Due to Gravity Below and Above the Earth's Surface
- Acceleration Due to Gravity and Its Variation with Altitude and Depth
- Gravitational Field
- Gravitational Potential Energy
- Escape Speed
- Earth Satellites
- Energy of an Orbiting Satellite
- Geostationary and Polar Satellites
- Weightlessness
- Escape Velocity
- Orbital Velocity of a Satellite
- Motion of Satellites
Properties of Solids and Liquids
- Introduction of Properties of Solids and Liquids
- Elastic Behaviour of Solid
- Stress and Strain
- Hooke’s Law
- Stress-strain Curve
- Elastic Moduli
- Young’s Modulus
- Determination of Young’s Modulus of the Material of a Wire
- Shear Modulus or Modulus of Rigidity
- Bulk Modulus
- Poisson’s Ratio
- Elastic Potential Energy in a Stretched Wire
- Application of Elastic Behaviour of Materials
- Pressure
- Pascal’s Law
- Variation of Pressure with Depth
- Atmospheric Pressure and Gauge Pressure
- Hydraulic Machines
- Streamline and Turbulent Flow
- Bernoulli's Equation
- Applications of Bernoulli’s Equation
- Torricelli's Law
- Viscous Force or Viscosity
- Stoke's Law
- Surface Tension
- Surface Energy
- Surface Tension and Surface Energy
- Angle of Contact
- Drops and Bubbles
- Capillary Rise
- Detergents and Surface Tension
- Heat and Temperature
- Measurement of Temperature
- Ideal-gas Equation and Absolute Temperature
- Thermal Expansion
- Specific Heat Capacity
- Calorimetry
- Change of State - Latent Heat Capacity
- Heat Transfer
- Conduction
- Convection
- Radiation
- Blackbody Radiation
- Qualitative Ideas of Black Body Radiation
- Wien's Displacement Law
- Green House Effect
- Newton’s Law of Cooling
- Reynold's Number
- Work Done in Stretching a Wire
- Terminal Velocity
- Capillarity and Capillary Action
- Fluid Flow
- Thermometer and Its Types
Thermodynamics
- Introduction of Thermodynamics
- Thermal Equilibrium
- Zeroth Law of Thermodynamics
- Heat, Internal Energy and Work
- First Law of Thermodynamics
- Specific Heat Capacity
- Thermodynamic State Variables and Equation of State
- Thermodynamic Process
- Isothermal Processes
- Adiabatic Processes
- Heat Engine
- Refrigerators and Heat Pumps
- Second Law of Thermodynamics
- Reversible and Irreversible Processes
- Carnot Engine
Kinetic Theory of Gases
- Introduction of Kinetic Theory of Gases
- Molecular Nature of Matter
- Gases and Its Characteristics
- Kinetic Theory of an Ideal Gas
- Law of Equipartition of Energy
- Specific Heat Capacities - Gases
- Mean Free Path
- Equation of State of a Perfect Gas
- Work Done in Compressing a Gas
- Interpretation of Temperature in Kinetic Theory
- Kinetic Theory of Gases - Concept of Pressure
- Assumptions of Kinetic Theory of Gases
- RMS Speed of Gas Molecules
- Degrees of Freedom
- Avogadro's Number
- The Gas Laws
- Kinetic Energy (K)
- Speed of Gas
Oscillations and Waves
- Introduction of Oscillations
- Periodic and Oscillatory Motion
- Simple Harmonic Motion (S.H.M.)
- Simple Harmonic Motion and Uniform Circular Motion
- Velocity and Acceleration in Simple Harmonic Motion
- Force Law for Simple Harmonic Motion
- Energy in Simple Harmonic Motion
- Some Systems Executing Simple Harmonic Motion
- Damped Simple Harmonic Motion
- Forced Oscillations and Resonance
- Displacement as a Function of Time
- Periodic Functions
- Oscillations - Frequency
- Wave Motion
- Transverse Waves and Longitudinal Waves
- Displacement Relation for a Progressive Wave
- The Speed of a Travelling Wave
- Principle of Superposition of Waves
- Reflection of Waves
- Introduction of Reflection of Waves
- Standing Waves and Normal Modes
- Beats
- Doppler Effect
- Interference
Electrostatics
- Introduction to Electrostatics
- Electric Charges
- Conductors and Insulators
- Charging by Induction
- Basic Properties of Electric Charge
- Coulomb’s Law - Force Between Two Point Charges
- Superposition Principle of Forces
- Superposition Principle - Forces Between Multiple Charges
- Electric Field
- Electric Field Due to a System of Charges
- Physical Significance of Electric Field
- Electric Field Lines
- Electric Flux
- Electric Dipole
- Dipole in a Uniform External Field
- Continuous Distribution of Charges
- Gauss’s Law
- Applications of Gauss’s Law
- Electric Field Due to a Point Charge
- Uniformly Charged Infinite Plane Sheet and Uniformly Charged Thin Spherical Shell (Field Inside and Outside)
- Electrostatic Potential
- Potential Due to a Point Charge
- Potential Due to an Electric Dipole
- Potential Due to a System of Charges
- Equipotential Surfaces
- Relation Between Electric Field and Electrostatic Potential
- Potential Energy of a System of Charges
- Potential Energy in an External Field
- Potential Energy of a Single Charge
- Potential Energy of a System of Two Charges in an External Field
- Potential Energy of a Dipole in an External Field
- Electrostatics of Conductors
- Dielectrics and Polarisation
- Capacitors and Capacitance
- The Parallel Plate Capacitor
- Effect of Dielectric on Capacity
- Combination of Capacitors
- Energy Stored in a Capacitor
- Electric Potential Energy
- Work Done in Carrying a Charge
- Grouping of Capacitor
Current Electricity
- Electric Current
- Electric Currents in Conductors
- Ohm's Law (V = IR)
- Drift of Electrons and the Origin of Resistivity
- Limitations of Ohm’s Law
- Resistivity of Various Materials
- Temperature Dependence of Resistance
- Electrical Energy
- Electrical Power
- Combination of Resistors - Series and Parallel
- Cells, Emf, Internal Resistance
- Kirchhoff’s Rules
- Wheatstone Bridge
- Metre Bridge
- Potentiometer
- V-I Characteristics (Linear and Non-linear)
- Electrical Resistivity and Conductivity
- Resistivity of Various Materials
- Combination of Cells in Series and in Parallel
- Heating Effect of Electric Current
- Cells, Thermo e.m.f. Electrolysis
Magnetic Effects of Current and Magnetism
- Magnetic Force
- Motion in a Magnetic Field
- Motion in Combined Electric and Magnetic Fields
- Velocity Selector
- Cyclotron
- Magnetic Field Due to a Current Element, Biot-Savart Law
- Magnetic Field on the Axis of a Circular Current Loop
- Ampere’s Circuital Law
- Solenoid and the Toroid - the Toroid
- Solenoid and the Toroid - the Solenoid
- Force Between Two Parallel Currents, the Ampere
- Torque on a Current Loop in Magnetic Field
- Moving Coil Galvanometer
- Force on a Current - Carrying Conductor in a Uniform Magnetic Field
- Force on a Moving Charge in Uniform Magnetic and Electric Fields
- Current Loop as a Magnetic Dipole and Its Magnetic Dipole Moment
- The Bar Magnet
- Magnetism and Gauss’s Law
- The Earth’s Magnetism
- Magnetisation and Magnetic Intensity
- Magnetic Properties of Materials
- Permanent Magnet and Electromagnet
- Hysteresis Loop
- Force and Torque on Current Carrying Conductor
- Properties of magnetic lines of force
- Magnetic Moment
- Magnetic Properties
- Magnetic Equipment
Electromagnetic Induction and Alternating Currents
- Electromagnetic Induction
- The Experiments of Faraday and Henry
- Magnetic Flux
- Faraday’s Law of Induction
- Lenz’s Law and Conservation of Energy
- Motional Electromotive Force (e.m.f.)
- Energy Consideration: a Quantitative Study
- Eddy Currents
- Inductance
- Mutual Inductance
- Self Inductance
- A.C. Generator
- Different Types of AC Circuits: AC Voltage Applied to a Resistor
- Representation of AC Current and Voltage by Rotating Vectors - Phasors
- Different Types of AC Circuits: AC Voltage Applied to an Inductor
- Different Types of AC Circuits: AC Voltage Applied to a Capacitor
- Different Types of AC Circuits: AC Voltage Applied to a Series LCR Circuit
- Power in AC Circuit: the Power Factor
- LC Oscillations
- Transformers
- Peak and Rms Value of Alternating Current Or Voltage
- Reactance and Impedance
- Alternating Currents
- Power in AC Circuit: the Power Factor
- Motional and Static EMI and Application of EMI
- Voltage and Power
- AC Circuits
- LCR Circuit
- Quality and Power Factor
Electromagnetic Waves
- Introduction of Electromagnetic Waves
- Displacement Current
- Electromagnetic Waves
- Electromagnetic Spectrum
- Applications of e.m. waves
- Transverse Nature of Electromagnetic Waves
Optics
- Reflection of Light by Spherical Mirrors
- Refraction
- Total Internal Reflection
- Refraction at Spherical Surfaces and Lenses
- Refraction at Spherical Surfaces
- Refraction by a Lens
- Combination of Thin Lenses in Contact
- Power of a Lens
- Refraction Through a Prism
- Some Natural Phenomena Due to Sunlight
- Optical Instruments
- Optical Instruments: Simple Microscope
- Optical Instruments: Compound Microscope
- Optical Instruments: Telescope
- Huygens' Principle
- Reflection and Refraction of Plane Wave at Plane Surface Using Huygens' Principle
- Refraction of a Plane Wave
- Refraction at a Rarer Medium
- Reflection of a Plane Wave by a Plane Surface
- The Doppler Effect
- Coherent and Incoherent Addition of Waves
- Interference of Light Waves and Young’s Experiment
- Diffraction of Light
- Fraunhofer Diffraction Due to a Single Slit
- Polarisation
- Ray Optics - Mirror Formula
- Dispersion of Light Through Prism and Formation of Spectrum
- Young's Double Slit Experiment and Expression for Fringe Width
- Coherent and Incoherent Sources and Sustained Interference of Light
- Resolving Power of Microscope and Astronomical Telescope
- Width of Central Maximum
- Lens Formula
- Magnification
- Brewster's Law
- Polaroids
- Plane Mirror
- Spherical Mirrors
- Reflection of Light
- Total Internal Reflection
- Power of a Lens
- Dispersion of Light Through Prism and Formation of Spectrum
- Resolving Power
Dual Nature of Matter and Radiation
- Dual Nature of Radiation
- Electron Emission
- The Photoelectric Effect
- Photoelectric Effect - Hertz’s Observations
- Photoelectric Effect - Hallwachs’ and Lenard’s Observations
- Experimental Study of Photoelectric Effect
- Photoelectric Effect and Wave Theory of Light
- Einstein’s Photoelectric Equation: Energy Quantum of Radiation
- Particle Nature of Light: The Photon
- Wave Nature of Matter
- Davisson and Germer Experiment
- Dual Behaviour of Matter: De Broglie's relationship
- Cathode and Positive Rays
- Photoelectric Effect X-rays
Atoms and Nuclei
- Introduction of Atoms
- Alpha-particle Scattering and Rutherford’s Nuclear Model of Atom
- Atomic Spectra
- Bohr’s Model for Hydrogen Atom
- Energy Levels
- The Line Spectra of the Hydrogen Atom
- De Broglie’s Explanation of Bohr’s Second Postulate of Quantisation
- Hydrogen Spectrum
- Atomic Masses and Composition of Nucleus
- Size of the Nucleus
- Mass-energy and Nuclear Binding Energy
- Mass - Energy
- Nuclear Binding Energy
- Atomic Mass, Mass - Energy Relation and Mass Defect
- Nuclear Force
- Radioactivity
- Introduction of Radioactivity
- Law of Radioactive Decay
- Alpha Decay
- Beta Decay
- Gamma Decay
- Nuclear Energy
- Introduction of Nuclear Energy
- Nuclear Fission
- Nuclear Reactor
- Nuclear Fusion – Energy Generation in Stars
- Controlled Thermonuclear Fusion
- Structure of an Atom
- Mass-Energy Equivalence and Nuclear Reactions
Electronic Devices
- Introduction to Semiconductors
- Classification of Metals, Conductors and Semiconductors
- Intrinsic Semiconductor
- Extrinsic Semiconductor
- p-n Junction
- Semiconductor Diode
- Application of Junction Diode as a Rectifier
- Special Purpose P-n Junction Diodes
- Zener Diode as a Voltage Regulator
- Digital Electronics and Logic Gates
- Junction Transistor
- Transistor Action
- Transistor and Characteristics of a Transistor
- Transistor as an Amplifier (Ce-configuration)
- Feedback Amplifier and Transistor Oscillator
Communication Systems
- Communication Systems
- Propagation of Electromagnetic Waves
- Need for Modulation and Demodulation
- Modulation and Its Necessity
- Amplitude Modulation (AM)
- Detection of Amplitude Modulated Wave
- Production of Amplitude Modulated Wave
- Frequency Modulation (FM)
- Bandwidth of Signals
- Bandwidth of Transmission Medium
- Elements of a Communication System
- Basic Terminology Used in Electronic Communication Systems
- Satellite Communication
Atomic Structure:
An atom consists of the nucleus and the extranuclear part, containing three types of subatomic particles: protons, neutrons, and electrons.
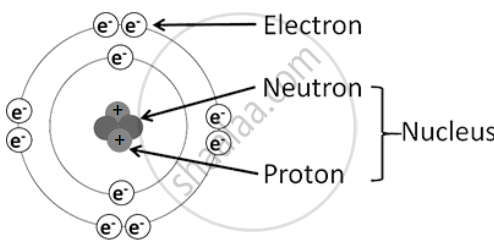
1. Nucleus: The nucleus is dense, positively charged, and contains almost all the atom’s mass. The particles inside the nucleus are protons and neutrons, collectively called nucleons.
2. Proton (p)
| Positive Charge | Protons are positively charged particles in the nucleus, symbolised as ‘p.’ |
| Charge and Symbol | Each proton is charged with +1e, where 1e = 1.6 × 10⁻¹⁹ coulomb. |
| Atomic Number (Z) | The number of protons in the nucleus defines the atomic number, denoted as ‘Z.’ |
| Mass | The mass of one proton is approximately 1 u (1 Dalton), about 1.66 × 10⁻²⁷ kg. |
|
Hydrogen Proton |
The mass of a hydrogen atom is also roughly 1 u. |
3. Neutron (n)
| Neutral Particle | Neutrons have no charge and are symbolised as ‘n.’ |
| Symbol for Number | The number of neutrons is denoted by ‘N.’ |
| Mass | The mass of a neutron is about 1 u, nearly equal to that of a proton. |
| Presence in Nuclei | All nuclei, except hydrogen with an atomic mass of 1u, contain neutrons. |
4. Extranuclear Part: This part consists of electrons moving around the nucleus and the empty space between them.
5. Electron (e⁻)
| Negative Charge | Electrons are negatively charged particles, symbolised as ‘e⁻.’ |
| Charge | Each electron is charged with -1e. |
| Mass | The mass of an electron is approximately 1/1800th of a hydrogen atom’s mass, negligible. |
| Electron Orbits | Electrons revolve in specific orbits, called shells, around the nucleus. |
| Shells | These orbits are three-dimensional, hence referred to as ‘shells.’ |
| Energy of Electrons | The energy of an electron depends on the shell it occupies. |
| Charge Balance | The number of electrons equals the number of protons (Z), making the atom electrically neutral. |
If you would like to contribute notes or other learning material, please submit them using the button below.
