Topics
The Language of Chemistry
- Matter (Substance)
- Molecules
- Pure Substances
- Elements
- Classification of Molecules
- Symbols Used to Represent Atoms of Different Elements
- Valency
- Variable Valency
- Chemical Formula or Molecular Formula
- Ions (Radicals) and Its Types
- Chemical Formula or Molecular Formula
- Naming Chemical Compounds
- To Calculate the Valency from the Formula
- Chemical Equation
- Balancing Chemical Equation
- Atomic Mass
- Molecular Mass
- Percentage Composition, Empirical and Molecular Formula
- Empirical Formula of a Compound
- Hydrogen
Chemical Changes and Reactions
Water
- Water: Our Lifeline
- Physical Properties of Water
- Chemical Properties of Water
- Water - a Universal Solvent
- Solutions as 'Mixtures' of Solids in Water
- Components of Solutions
- Different Types of Solutions
- Saturated Solutions
- Concentration of a Solution
- Solubility
- Crystals and Crystallisation
- Hydrated and Anhydrous Substances
- Efflorescence, Hygroscopic, and Deliquescence Substances
- Drying and Dehydrous Substances
- Classification of water: Soft and Hard Water
- Causes of Hardness
- Advantage and Disadvantage of Hard Water
- Removal of Hardness of Water
- Salts
- Prevention of Water Pollution
Atomic Structure and Chemical Bonding
- Chemical Bond
- History of Atom
- Dalton’s Atomic Theory
- Elements
- Atoms: Building Blocks of Matter
- Discovery of Charged Particles in Matter
- Electrons (e)
- Protons (p)
- Nucleus
- Neutrons (n)
- J. J. Thomson’s Atomic Model
- Lord Rutherford’s Atomic model
- Neils Bohr’s Model of an Atom
- Structure of an Atom
- Atomic Number (Z), Mass Number (A), and Number of Neutrons (n)
- Atomic Mass
- Electronic Configuration of Atom
- Valency
- Reason for Chemical Activity of an Atom
- Isotopes
- Isobars
- Ionic or Electrovalent Bond
- Ionic or Electrovalent Bond
- The Covalent Bond
- Types of Covalent Bond
- Formation of Covalent Bond
The Periodic Table
- History of Periodic Table: Early Attempts at the Classification of Elements
- Dobereiner’s Triads
- Newland's Law of Octaves
- Mendeleev’s Periodic Table
- Merits and Demerits of Mendeleev’s Periodic Table
- Modern Periodic Law
- The Modern Periodic Table
- Structure of the Modern Periodic Table
- Advantage and Disadvantage of Modern Periodic Table
- Periodic Properties
- Shells (Orbits)
- Valency
- Properties of Elements
- Atomic Radius Or Atomic Size
- Metallic and Non-metallic Characters
- Study of Specific Groups in Periodic Table
- Group I (Alkali Metals)
- Group II (Alkaline Earth Metals)
- Group VIIA Or Group 17 (The Halogens)
- Group Zero or 18 Group (Noble Gases)
- Uses of Periodic Table
- Types of Element: Metals
Study of the First Element - Hydrogen
- Position of Hydrogen in Periodic Table
- Similarities Between Hydrogen and Alkali Metals
- Similarities Between Hydrogen and Halogens
- Hydrogen
- Preparation of Hydrogen
- Application of Activity Series in the Preparation of Hydrogen
- Laboratory Preparation of Hydrogen
- Manufacture of Hydrogen
- Physical Properties of Hydrogen
- Chemical Properties of Hydrogen
- Uses of Hydrogen
- Oxidation, Reduction and Redox Reactions
Study of Gas Laws
- Gases and Its Characteristics
- Molecular Motion : Relationship of Temperature, Pressure and Volume
- The Gas Laws
- Pressure and Volume Relationship or Bolye's Law
- Temperature - Volume Relationship or Charles's Law
- Absolute Zero
- The Temperature and a Thermometer
- Scales of Thermometers
- Gas Equation
- Standard Temperature Pressure (S.T.P.)
- The Effect of Moisture and Pressure
Atmospheric Pollution
- Atmospheric Pollution
- Air Pollution and Its Causes
- Effects of Air Pollution
- Prevention of Air Pollution
- Gaseous Pollutants and Their Effects
- Acid Rain
- Causes of Acid Rain
- Green House Effect
- Advantage of Green House Effect
- Global Warming
- Preventive Measures of Global Warming
- Ozone
- Ozone Layer Depletion
Elements, Compounds and Mixtures
- Differences Between Elements, Compounds, and Mixtures
- Types of Mixtures
- Mixture
- Separation of Mixtures
- Use of Solvent and Filtration
- Concept of Evaporation
- Simple Distillation Method
- Simple Distillation Method
- Chromatography Method
- Centrifugation Method
- Solvent Extraction (Using a Separating Funnel Method)
Matter and Its Composition: Law of Conservation of Mass
- Heat and change of physical state
- Inter-particle Space and Interparticle Attraction and Collision
- Law of Conservation of Mass
Practical Work
- Laboratory Preparation of Hydrogen
- Laboratory Preparation of Oxygen
- Laboratory Preparation of Carbon Dioxide
- Laboratory Preparation of Chlorine
- Laboratory Preparation of Hydrogen Chloride Gas
- Laboratory Preparation of Sulphur Dioxide
- Laboratory Preparation of Hydrogen Sulphide
- Laboratory Preparation of Ammonia Gas
- Laboratory Preparation of Water Vapour
- Laboratory Preparation of Nitrogen Dioxide
- Action of Heat on a Given Substance
- Action of Dilute Sulphuric Acid on a Given Substance
- Dry Test
- Recognition of Substances by Colour
- Recognition of Substances by Odour
- Recognition of Substances by Physical State
- Recognition of Substances by Action of Heat
- Flame Test
- Classification of water: Soft and Hard Water
- Simple Experiments Based on Hard Water and Soft Water
- Water Pollution and Its Causes
- Water Quality
- Strength of Acidic or Basic Solutions
- Prevention of Water Pollution
Atomic Structure:
An atom consists of the nucleus and the extranuclear part, containing three types of subatomic particles: protons, neutrons, and electrons.
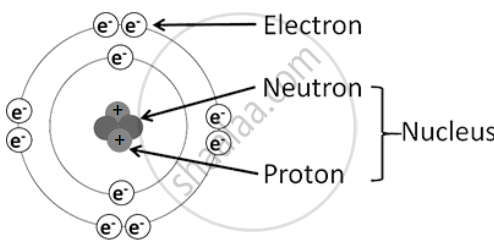
1. Nucleus: The nucleus is dense, positively charged, and contains almost all the atom’s mass. The particles inside the nucleus are protons and neutrons, collectively called nucleons.
2. Proton (p)
| Positive Charge | Protons are positively charged particles in the nucleus, symbolised as ‘p.’ |
| Charge and Symbol | Each proton is charged with +1e, where 1e = 1.6 × 10⁻¹⁹ coulomb. |
| Atomic Number (Z) | The number of protons in the nucleus defines the atomic number, denoted as ‘Z.’ |
| Mass | The mass of one proton is approximately 1 u (1 Dalton), about 1.66 × 10⁻²⁷ kg. |
|
Hydrogen Proton |
The mass of a hydrogen atom is also roughly 1 u. |
3. Neutron (n)
| Neutral Particle | Neutrons have no charge and are symbolised as ‘n.’ |
| Symbol for Number | The number of neutrons is denoted by ‘N.’ |
| Mass | The mass of a neutron is about 1 u, nearly equal to that of a proton. |
| Presence in Nuclei | All nuclei, except hydrogen with an atomic mass of 1u, contain neutrons. |
4. Extranuclear Part: This part consists of electrons moving around the nucleus and the empty space between them.
5. Electron (e⁻)
| Negative Charge | Electrons are negatively charged particles, symbolised as ‘e⁻.’ |
| Charge | Each electron is charged with -1e. |
| Mass | The mass of an electron is approximately 1/1800th of a hydrogen atom’s mass, negligible. |
| Electron Orbits | Electrons revolve in specific orbits, called shells, around the nucleus. |
| Shells | These orbits are three-dimensional, hence referred to as ‘shells.’ |
| Energy of Electrons | The energy of an electron depends on the shell it occupies. |
| Charge Balance | The number of electrons equals the number of protons (Z), making the atom electrically neutral. |
If you would like to contribute notes or other learning material, please submit them using the button below.
