Advertisements
Advertisements
Write the structures of the main products in the following reactions :
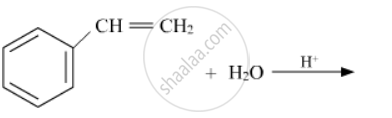
Concept: Introduction to Haloalkanes and Haloarenes > Nomenclature
What happens when: XeF4 reacts with SbF5?
Concept: P - Block Group 18 Elements > Concept of Group 18 Elements
Out of chlorobenzene and benzyl chloride, which one gets easily hydrolysed by aqueous NaOH and why?
Concept: Compounds of Halogens
Write the IUPAC name of the following :
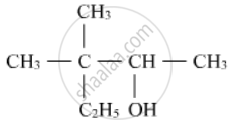
Concept: Alcohols, Phenols and Ethers > Nomenclature
Among the hydrides of Group-15 elements, which have the lowest boiling point?
Concept: Concept of Group 15 Elements
Among the hydrides of Group-15 elements, which have the maximum basic character?
Concept: Concept of Group 15 Elements
Among the hydrides of Group-15 elements, which have the highest bond angle?
Concept: Concept of Group 15 Elements
Among the hydrides of Group-15 elements, which have the maximum reducing character?
Concept: Concept of Group 15 Elements
Identify the chiral molecule in the following pair :
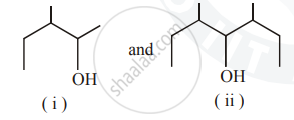
Concept: Physical and Chemical Properties of Alcohols and Phenols
Write the structure of the product when chlorobenzene is treated with methyl chloride in the presence of sodium metal and dry ether.
Concept: Compounds of Halogens
Write the structure of the alkene formed by dehydrohalogenation of 1-Bromo-1methylcyclohexane with alcoholic KOH.
Concept: Compounds of Halogens
Write the structures of the main products in the following reactions :
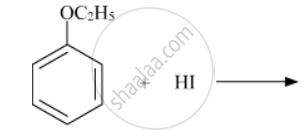
Concept: Chemical Reaction of Ethers - Cleavege of C-O Bonds
Give reasons H3PO3 undergoes disproportionation reaction but H3PO4 does not.
Concept: Concept of Group 15 Elements
Give reasons When Cl2 reacts with the excess of F2, ClF3 is formed and not FCl3.
Concept: Concept of Group 15 Elements
Give reasons Dioxygen is a gas while Sulphur is a solid at room temperature.
Concept: P - Block Group 16 Elements > Sulphur - Allotropic Forms
Propanal and Propanone
Concept: Aldehydes and Ketones > Chemical Reactions of Aldehydes and Ketones - Oxidation
Account for the following:
Aromatic carboxylic acids do not undergo Friedel-Crafts reaction.
Concept: Acids > Structure of the Carboxyl group
Give reason for the following:
Primary amines have higher boiling point than tertiary amines.
Concept: Physical Properties of Amines
Illustrate the following reaction giving suitable example in each case: Diazotisation
Concept: Diazonium Salts > Method of Preparation of Diazonium Salts
Give reason (CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Concept: Physical Properties of Amines
