Advertisements
Advertisements
प्रश्न
Give reasons H3PO3 undergoes disproportionation reaction but H3PO4 does not.
उत्तर
In H3PO3, P is present in +3 oxidation state while in H3PO4, P is present in +5 oxidation state. Since, in H3PO4, P is present in the maximum oxidation state, so it does not undergo disproportionation reaction. The reaction for disproportionation of H3PO3 is given as follows:
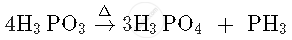
APPEARS IN
संबंधित प्रश्न
Why is BiH3 the strongest reducing agent amongst all the hydrides of Group 15 elements?
Maximum covalency of nitrogen is ______.
Which one of the following elements is most metallic?
With respect to protonic acids, which of the following statement is correct?
H2S is more acidic than H2O because ____________.
The oxidation state of central atom in the anion of compound \[\ce{NaH2PO2}\] will be ______.
Which of the following options are not in accordance with the property mentioned against them?
| (i) | \[\ce{F2 > Cl2 > Br2 > I2}\] | Oxidising power. |
| (ii) | \[\ce{MI > MBr > MC1 > MF}\] | Ionic character of metal halide. |
| (iii) | \[\ce{F2 > Cl2 > Br2 > I2}\] | Bond dissociation enthalpy. |
| (iv) | \[\ce{HI < HBr < HCI < HF}\] | Hydrogen-halogen bond strength. |
Which of the following pairs will have same chemical properties?
The correct order of increase acidic strength of CIOH(1), BrOH (II) and lOH (iii) is.
Chemical nature of the nitrogen oxide compound obtained from a reaction of concentrated nitric acid and P4O10 (in 4 : 1 ratio) is ______.
