Advertisements
Advertisements
प्रश्न
Give reasons H3PO3 undergoes disproportionation reaction but H3PO4 does not.
उत्तर
In H3PO3, P is present in +3 oxidation state while in H3PO4, P is present in +5 oxidation state. Since, in H3PO4, P is present in the maximum oxidation state, so it does not undergo disproportionation reaction. The reaction for disproportionation of H3PO3 is given as follows:
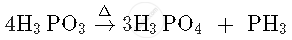
APPEARS IN
संबंधित प्रश्न
Write four points of differences between properties of nitrogen and other elements of group 15.
The element that does NOT exhibit allotropy is
- As
- Sb
- Bi
- N
Arrange the following in the increasing order of property mentioned :
NH3, PH3, AsH3, SbH3, BiH3 (Base strength)
[Ar]3d104s24p3 is the electronic configuration of ____________.
The strongest oxidising agent is ____________.
Which of the following has the highest pπ–pπ bonding tendency?
Which of the following statements are correct for \[\ce{SO2}\] gas?
(i) It acts as bleaching agent in moist conditions.
(ii) It’s molecule has linear geometry.
(iii) It’s dilute solution is used as disinfectant.
(iv) It can be prepared by the reaction of dilute \[\ce{H2SO4}\] with metal sulphide.
Match the species given in Column I with the shape given in Column II and mark the correct option.
| Column I | Column II |
| (A) \[\ce{SF4}\] | (1) Tetrahedral |
| (B) \[\ce{BrF2}\] | (2) Pyramidal |
| (C) \[\ce{BrO^{-}3}\] | (3) Sea-saw shaped |
| (D) \[\ce{NH^{+}4}\] | (4) Bent T-shaped |
Assertion: N2 is less reactive than P4.
Reason: Nitrogen has more electron gain enthalpy than phosphorus.
Which acid of the following pair would you expect to be stronger?
F−CH2−COOH or CH3−COOH
