Advertisements
Advertisements
Question
Give reasons H3PO3 undergoes disproportionation reaction but H3PO4 does not.
Solution
In H3PO3, P is present in +3 oxidation state while in H3PO4, P is present in +5 oxidation state. Since, in H3PO4, P is present in the maximum oxidation state, so it does not undergo disproportionation reaction. The reaction for disproportionation of H3PO3 is given as follows:
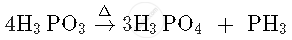
APPEARS IN
RELATED QUESTIONS
Why are pentahalides more covalent than trihalides?
Account for the following :
N − N single bond is weaker than P − P single bond.
Account for the following :
Sulphur in vapour form exhibits paramagnetic behaviour.
The correct decreasing order of basic strength is:
The ion or group detected by K2[HgI4] is ____________.
Acidity of diprotic acid in aqueous solution increases in order.
Hess's law is used to calculate:
Among the following, identify the species with an atom in +6 oxidation state
Basicity of ortho phosphoric acid is:-
Chemical nature of the nitrogen oxide compound obtained from a reaction of concentrated nitric acid and P4O10 (in 4 : 1 ratio) is ______.
