Advertisements
Advertisements
प्रश्न
Propanal and Propanone
उत्तर १
Propanal and propanone can be distinguished by the following tests:
- Tollen’s test: Propanal is an aldehyde. Thus, it reduces Tollen’s reagent. But, propanone being a ketone does not reduce Tollen’s reagent.
\[\ce{\underset{Propanal}{CH3CH2CHO} + \underset{Tollen's reagent}{2[Ag(NH3)2]+} + 3OH- -> \underset{Propanoate ion}{CH3CH2COO-} + \underset{Silver mirror}{Ag\downarrow} + NH3 + 2H2O}\] - Fehling’s test: Aldehydes respond to Fehling’s test, but ketones do not. Propanal being an aldehyde reduces Fehling’s solution to a red-brown precipitate of Cu2O, but propanone being a ketone does not.
\[\ce{\underset{Propanal}{CH3CH2CHO} + 2Cu^2+ + 5OH- -> \underset{Propanaoate ion}{CH3CH2COO-} + \underset{(Red-brown ppt)}{\underset{Cuprous oxide}{Cu2O\downarrow}}+ 2H2O}\] - Iodoform test: Aldehydes and ketones with at least one methyl group linked to the carbonyl carbon atom respond to the iodoform test. They are oxidized by sodium hypoiodite (NaOI) to give iodoforms. Propanone being a methyl ketone responds to this test, but propanal does not.
\[\ce{\underset{Propanone}{CH3COCH3} + 3NaOI -> \underset{Sodium acetate}{CH3COONa} + \underset{(yellow ppt)}{\underset{Iodoform}{CHI3}}+ 2NaOH}\]
उत्तर २
| Reagent | Propanal | Propanone |
| Tollen’s Reagent | On heating with Tollen’s reagent, a silver mirror is formed on the inner walls of the test tube. Aldehyde groups reduce the Tollen's reagent. | No change in propanone. |
APPEARS IN
संबंधित प्रश्न
Give a simple chemical test to distinguish between the following pair of compounds:
Ethanal and Propanal
Give a simple chemical test to distinguish between the following pair of compounds:
Pentan-2-one and Pentan-3-one
Alkenes decolourise bromine water in presence of CCl4 due to formation of ______.
Oxidation of ketones involves carbon-carbon bond cleavage. Name the products formed on oxidation of 2, 5-dimethylhexan-3-one.
Solvent used for dewaxing of petroleum products are
Acetaldehyde cannot show?
Ammonical silver nitrate solution is called
A hydrocarbon (A) with molecular formula C5H10 on ozonolysis gives two products (B) and (C). Both (B) and (C) give a yellow precipitate when heated with iodine in presence of NaOH while only (B) give a silver mirror on reaction with Tollen’s reagent.
- Identify (A), (B) and (C).
- Write the reaction of B with Tollen’s reagent.
- Write the equation for iodoform test for C.
- Write down the equation for aldol condensation reaction of B and C.
Write chemical test to distinguish between the following compounds:
Phenol and Benzoic acid
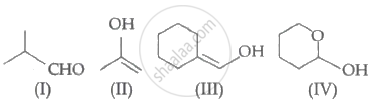
Which among the above compound/s does/do not form Silver mirror when treated with Tollen's reagent?
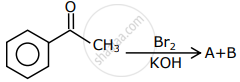
The major products formed in the following reaction sequence A and B are:
An organic compound neither reacts with neutral ferric chloride solution nor with Fehling solution. It however, reacts with Grignard reagent and gives positive iodoform test. The compound is:
The correct set of products obtained in the following reactions:
- \[\ce{RCN ->[reduction]}\]
- \[\ce{RCN ->[(i) CH3MgBr][(ii) H2O]}\]
- \[\ce{RNC ->[hydrolysis]}\]
- \[\ce{RNH2 ->[HNO2]}\]
Choose the reaction which is not possible:
In Tollen's test for aldehyde, the overall number of electrons(s) transferred to the Tollen's reagent formula \[\ce{[Ag(NH3)2]+}\] per aldehyde group to form silver mirror is ______. (Round off to the nearest integer)
The reagent that can be used to distinguish acetophenone and benzophenone is ______.
An organic compound 'A' with molecular formula C5H8O2 is reduced to n-pentane with hydrazine followed by heating with NaOH and glycol. 'A' forms a dioxime with hydroxylamine and gives a positive iodoform and Tollen's test. Identify 'A' and give its reaction for iodoform and Tollen's test.
