Advertisements
Advertisements
प्रश्न
10g of ice at 0℃ absorbs 5460 J of heat energy to melt and change to water at 50℃. Calculate the specific latent heat of fusion of ice. Specific heat capacity of water is 4200 J kg-1 K-1.
उत्तर
Given, Mass of ice = 10g
Heat energy absorbed (Q) = 5460 J
Specific heat capacity of water = 4200 J kg-1 K-1 = 4.2 J g-1 K-1
Specific latent heat of fusion of ice = ?
(i) Heat energy required to melt the ice at 0° C to water at 0° C (Q1) = m × L
Substituting the values in the formula we get,
Q1 = 10 × L
(ii) Heat energy required to raise temperature from 0° C to 50° C = m × c × rise in temperature
Substituting the values in the formula we get,
Q2 = 10 × 4.2 × 50
Q2 = 2100 J
Then,
Q = Q1 + Q2
∴ 5460 J = 10 × L + 2100 J
∴ 5460 J - 2100 J = 10 × L
∴ 3360 = 10 × L
∴ `3360/10` = L
∴ L = 336 Jg-1
APPEARS IN
संबंधित प्रश्न
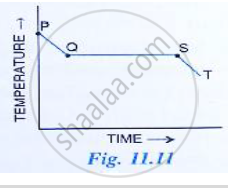
Fig 11. 11 shows the variation in temperature with time when some wax cools from the liquid phase to the solid phase.
(i) In which part of the curve, the wax is in liquid phase?
(ii) What does the part QS of the curve represent?
(iii) In which part of the curve, the wax will be the in the liquid as well as solid phase?
(iv) In which part of the curve, the wax is in solid phase?
(i) Name the physical quantity which is measured in calories.
(ii) How is calorie related to the S.I. unit of that quantity ?
Express -400C on the Fahrenheit scale.
State the principle of calorimeter and define calorie and kilocalorie.
Fill in the following blank using suitable word:
Whenever mechanical work is done, .......... Is produced.
Name two factors on which the heat energy librated by a body on cooling depends.
Explain, why is water used as a coolent in motor car radiators?
A copper calorimeter of mass 50g contains 100g of water at 20°C. A metallic piece of mass 250 g is heated to 100°C and is then dropped into the calorimeter. The contents of the calorimeter are well stirred and its final highest temperature is recorded to be 28 °C. If the specific heat capacity of water is 4.2 J/g°C and of copper is 0.4 J/g°C, find:
(i) the heat gained by water,
(ii) the heat gained by calorimeter,
(iii) total heat supplied by the metal piece, and
(iv) the specific heat capacity of metal.
Find the final temperature when a mass of 80g of water at 100°C is mixed with a mass of 40g of water at 25°C.
