Advertisements
Advertisements
प्रश्न
A calorimeter is kept in a wooden box to insulate it thermally from the surroundings. Why is it necessary?
उत्तर
A calorimeter is kept in a wooden box to insulate it thermally from the surroundings because in order to determine the specific heat capacity, the total heat transferred must be known. Heat must not be exchanged with the surroundings, otherwise the principle of calorimeter would not stand valid.
APPEARS IN
संबंधित प्रश्न
The calorie is defined as 1 cal = 4.186 joule. Why not as 1 cal = 4 J to make the conversions easy?
Three bodies of the same material and having masses of m, m and 3m are at temperatures of 40°C, 50°C and 60°C respectively. If the bodies are brought in thermal contact, the final temperature will be:
Refer to the plot of temperature versus time (figure) showing the changes in the state of ice on heating (not to scale).
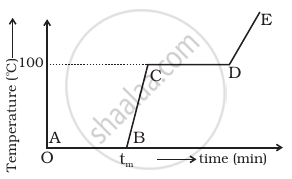
- The region AB represents ice and water in thermal equilibrium.
- At B water starts boiling.
- At C all the water gets converted into steam.
- C to D represents water and steam in equilibrium at boiling point.
On an X temperature scale, water freezes at -125°X and boils at 375°X. On a Y temperature scale, water freezes at -70°Y and boils at -30°Y. The value of temperature on X scale is ______ on which value of temperature on y Scale becomes 50° Y.
A copper block of mass 2.5 kg is heated in a furnace to a temperature of 500°C and then placed on large ice block. The maximum amount of ice that can melt is ______.
(Specific heat of copper = 0.39 Jg-1 c-1, latent heat of 0 fusion of water = 335 Jg-1)
80 gm of water at 30°C is poured on a large block of ice at 0°C. The mass of ice that melts is ______.
Work done in converting one gram of ice at -10°C into steam at 100°C is ______.
The specific heat of a substance is given by C = a + bT, where a = 1.12 kJ kg-1K-1 and b = 0.016 kJ-kg K-2. The amount of heat required to raise the temperature of 1.2 kg of the material from 280 K to 312 K is ______.
500 g of water and 100 g of ice at 0°C are in a calorimeter whose water equivalent is 40 g. 10 g of steam at 100°C is added to it. Then water in the calorimeter is ______.
(Latent heat of ice = 80 cal/g, Latent heat of steam = 540 cal/g)
540 g of ice at 0°C is mixed with 540 g of water at 80°C. The final temperature of the mixture will be ______.
