Advertisements
Advertisements
प्रश्न
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – zinc carbonate.
उत्तर
Change of colour — In certain chemical reactions a change of colour is seen to be observed. Heat on substances to observe change in colour of reactants and products.
Heat on:
Zinc carbonate ZnCO3 → ZnO + CO2 [g]
Colour change:
Zinc carbonate [ZnCO3] – White – to Zinc oxide [ZnO] – Yellow (when hot)
APPEARS IN
संबंधित प्रश्न
Correct and balance the following equation:
Ca + H2O  CaOH + H
CaOH + H
Giving examples, state the difference between balanced and unbalanced chemical equations.
Balance the following equation and add state symbols:
Zn + HCI → ZnCI2 + H2
A metal X forms a salt XSO4. The salt XSO4 forms a clear solution in water which reacts with sodium hydroxide solution to form a blue precipitate Y. Metal X is used in making electric wire and alloys like brass.
(a) What do you think metal X could be?
(b) Write the name, formula and colour of salt XSO4.
(c) What is the blue precipitate Y?
(d) Write a chemical equation of the reaction which takes place when salt XSO4 reacts with sodium hydroxide solution. Give the state symbols of all the reactants and products which occur in the above equation.
Balance the following chemical equation:
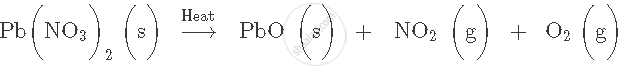
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Copper [II] sulphate to sodium hydroxide.
Representation of the results of a chemical change – is a chemical equation.
For the equation: FeCl3 + 3NH4OH 3NH4Cl + Fe(OH)3 ↓
Answer the following:
What is the indications of the arrow between the reactants and the products and of the arrow pointing downwards at the end.
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
\[\ce{N2 + H2 <=> NH3}\]
CaCO3 + 2HCl[dil.] → CaCl2 + H2O + CO2 [g]
State the information not conveyed by the above chemical equation.
