Advertisements
Advertisements
प्रश्न
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
उत्तर
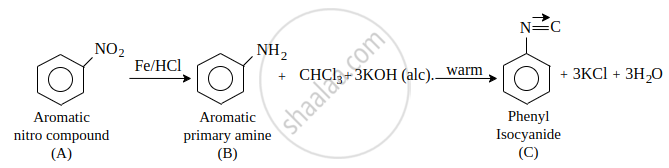
The compound A is Nitrobenzene, B is Aniline and C is Phenyl isocyanide.
संबंधित प्रश्न
Write the order of reactivity of alkyl halides with ammonia.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
