Advertisements
Advertisements
Question
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Solution
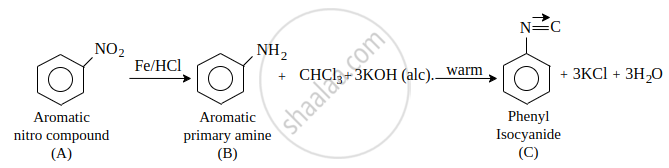
The compound A is Nitrobenzene, B is Aniline and C is Phenyl isocyanide.
RELATED QUESTIONS
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structures of A, B and C in the following reactions :
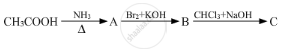
Write reactions to bring about the following conversions.
Acetamide to methylamine
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
Reduction of nitrobenzene by which of the following reagent gives aniline?
(i) \[\ce{Sn/HCl}\]
(ii) \[\ce{Fe/HCl}\]
(iii) \[\ce{H2 - Pd}\]
(iv) \[\ce{Sn/NH4OH}\]
When primary amines are treated with HCl, the product obtained is which of the following?
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Write a short note on the following:
Ammonolysis
