Advertisements
Advertisements
Question
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Solution
\[\ce{\underset{chloride}{\underset{Benzenediazonium}{C6H5N2Cl}} ->[CuCN] \underset{(A)}{\underset{Cyanobenzene}{C6H5CN}} ->[H2O/H+] \underset{(B)}{\underset{Benzoic acid}{C6H5COOH}} ->[NH3][\Delta] \underset{(C)}{\underset{Benzamide}{C6H5CONH2}}}\]
APPEARS IN
RELATED QUESTIONS
How is ethyl amine prepared from methyl iodide?
Give the structures of A, B and C in the following reaction:
\[\ce{CH3COOH ->[NH3][\Delta] A ->[NaOBr] B ->[NaNO2/HCl] C}\]
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Write structures of compounds A and B in each of the following reactions: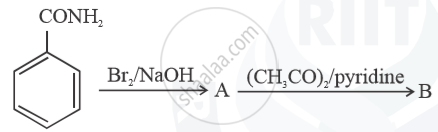
Write the order of reactivity of alkyl halides with ammonia.
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
Explain the following reaction with a suitable example.
Hofmann elimination reaction
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Which of the following compounds is the weakest Brönsted base?
Identify A and B in the following reaction.
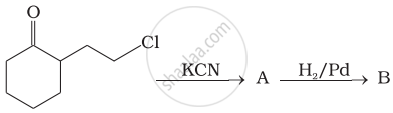
How will you carry out the following conversion?
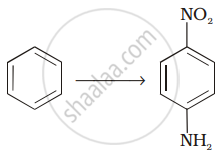
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
Reduction of nitro alkanes yields which compound?
Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Which of the following compound gives pink colour on reaction with phthalic anhydride in cone. H2SO4 followed by treatment with NaOH?
Amides can be converted into amines by the reaction named ______.
Write short note on the following:
Ammonolysis
