Advertisements
Advertisements
प्रश्न
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
उत्तर
\[\ce{\underset{chloride}{\underset{Benzenediazonium}{C6H5N2Cl}} ->[CuCN] \underset{(A)}{\underset{Cyanobenzene}{C6H5CN}} ->[H2O/H+] \underset{(B)}{\underset{Benzoic acid}{C6H5COOH}} ->[NH3][\Delta] \underset{(C)}{\underset{Benzamide}{C6H5CONH2}}}\]
APPEARS IN
संबंधित प्रश्न
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Give the structures of A, B and C in the following reaction:
\[\ce{C6H5NO2 ->[Fe/HCl] A ->[HNO2][273 K] B ->[C6H5OH] C}\]
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
____________ can be prepared exclusively by Gabriel phthalimide synthesis.
Which of the following amines forms a clear solution when treated with benzene sulphonyl chloride and excess of potassium hydroxide?
Identify the product obtained when benzamide is treated with bromine and aqueous sodium hydroxide?
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Suggest a route by which the following conversion can be accomplished.
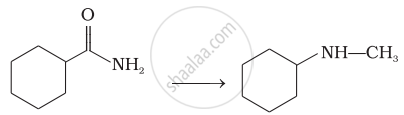
How will you carry out the following conversions?
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Acetamide and ethyl amide can be distinguished by reacting with.
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis
Write a short note on the following:
Ammonolysis.
Write a short note on the following:
Ammonolysis
