Advertisements
Advertisements
Question
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Solution
\[\begin{array}{cc}
\phantom{.......................}\ce{O}\\
\phantom{.......................}||\\
\ce{\underset{Iodoethane}{CH3CH2I} ->[NaCN] \underset{(A)}{\underset{Propane nitrile}{CH3CH2CN}} ->[OH-][Partial hydrolysis] \underset{(B)}{\underset{Propanamide}{CH3CH2-C-NH2}} ->[NaOH + Br2] \underset{(C)}{\underset{Ethanamine}{CH3CH2-NH2}}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Identify the compounds 'A' and 'B' in the following equation:

How do you convert the following: Ethanenitrile to ethanamine
Write a short note on the following:
Hoffmann’s bromamide reaction
Accomplish the following conversion:
Benzamide to toluene
Write the reaction of aliphatic primary amine with nitrous acid.
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Name the process of breaking C-X bond by ammonia in preparation of amines.
Acetamide on reduction using Na/C2H5OH gives ____________.
Quaternary ammonium salt is formed:
The best reagent for converting, 2-phenylpropanamide into 1- phenylethanamine is ______.
Reduction of aromatic nitro compounds using \[\ce{Fe}\] and \[\ce{HCl}\] gives ______.
Which of the following reactions are correct?
(i)
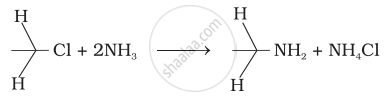
(ii)
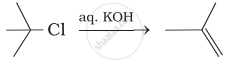
(iii)
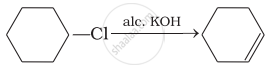
(iv)
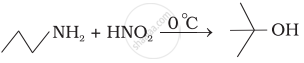
What is the product when \[\ce{C6H5CH2NH2}\] reacts with \[\ce{HNO2}\]?
How will you bring out the following conversion?

Match the reactions given in Column I with the statements given in Column II.
| Column I | Column II | ||
| (i) | Ammonolysis | (a) | Amine with lesser number of carbon atoms |
| (ii) | Gabriel phthalimide synthesis | (b) | Detection test for primary amines. |
| (iii) | Hoffmann Bromamide reaction | (c) | Reaction of phthalimide with \[\ce{KOH}\] and \[\ce{R-X}\] |
| (iv) | Carbylamine reaction | (d) | Reaction of alkylhalides with \[\ce{NH3}\] |
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
Assertion: Aromatic 1° amines can be prepared by Gabriel Phthalimide Synthesis.
Reason: Aryl halides undergo nucleophilic substitution with anion formed by phthalimide.
Acetamide and ethyl amide can be distinguished by reacting with.
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
