Advertisements
Advertisements
Question
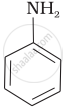
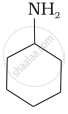
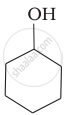
Which of the following compounds is the weakest Brönsted base?
Options
Solution

Explanation:
Amines (i, ii) have stronger tendency to accept a proton and hence are stronger Bronsted bases than phenol (iii) and alcohol (iv). Since phenol is more acidic than alcohol, therefore, phenol (iii) has the least tendency to accept a proton and hence it is the weakest Bronsted base.
APPEARS IN
RELATED QUESTIONS
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Give the structures of A, B and C in the following reactions :

Answer the following
Explain Gabriel phthalimide synthesis.
Quaternary ammonium salt is formed:
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Write following conversions:
acetanilide `->` p-nitroaniline
How will you carry out the following conversions?
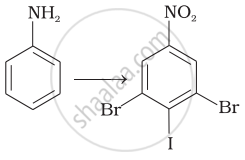
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Write short note on the following:
Ammonolysis
Write short notes on the following:
Ammonolysis