Advertisements
Advertisements
Question
Among the following amines, the strongest Brönsted base is:
Options
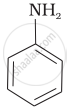
\[\ce{NH3}\]
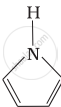
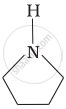
Solution

Explanation:
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
APPEARS IN
RELATED QUESTIONS
Write the reactions of aromatic with nitrous acid.
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :
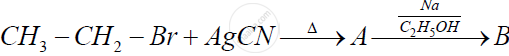
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Name the process of breaking C-X bond by ammonia in preparation of amines.
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Write the name of the product formed by the action of LiAlH4/ether on acetamide.
Write short note on the following.
Ammonolysis.
