Advertisements
Advertisements
प्रश्न
Among the following amines, the strongest Brönsted base is:
पर्याय
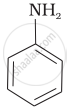
\[\ce{NH3}\]
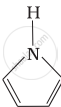
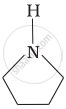
उत्तर

Explanation:
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
APPEARS IN
संबंधित प्रश्न
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.

Identify the major product (B).
What product is formed when \[\ce{R - C ≡ N}\] is hydrolysed?
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Acetamide and ethyl amide can be distinguished by reacting with.
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Amides can be converted into amines by the reaction named ______.
Write the name of reduction product formed when ethyl cyanide is treated with sodium and alcohol.
