Advertisements
Advertisements
प्रश्न
Among the following amines, the strongest Brönsted base is:
विकल्प
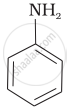
\[\ce{NH3}\]
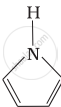
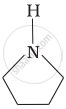
उत्तर

Explanation:
Pyrrolidine is the strongest of two bases because the lone pair of nitrogen does not involve sin resonance and the presence of two alkyl basic compounds increases the basic strength among the given four compounds.
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Answer the following
Explain Gabriel phthalimide synthesis.
Write reactions to prepare ethanamine from Acetonitrile.
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
How will you carry out the following conversion?
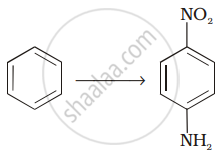
Account for the following:
Aniline cannot be prepared by the ammonolysis of chlorobenzene under normal conditions.
Which of the following compound is expected to be most basic?
Amides can be converted into amines by the reaction named ______.
