Advertisements
Advertisements
प्रश्न
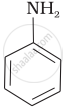
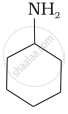
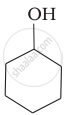
Which of the following compounds is the weakest Brönsted base?
विकल्प
उत्तर

Explanation:
Amines (i, ii) have stronger tendency to accept a proton and hence are stronger Bronsted bases than phenol (iii) and alcohol (iv). Since phenol is more acidic than alcohol, therefore, phenol (iii) has the least tendency to accept a proton and hence it is the weakest Bronsted base.
APPEARS IN
संबंधित प्रश्न
Illustrate the following reaction giving suitable example in each case:Gabriel phthalimide synthesis
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Choose the most correct option.
Which of the following compounds will dissolve in aqueous NaOH after undergoing reaction with Hinsberg reagent?
Write reactions to bring about the following conversions.
Acetamide to Ethylamine
Write reactions to bring about the following conversions.
Acetamide to methylamine
Identify the product obtained, when benzamide is treated with bromine and aqueous sodium hydroxide.
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
Identify 'A' and 'B' in the following conversions.
\[\ce{CH3 - I ->[Alc. KCN][\Delta] A ->[Na/C2H5OH] B}\]
Which of the following reactions are correct?
(i)
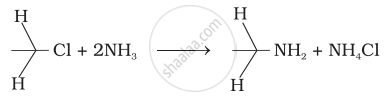
(ii)
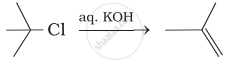
(iii)
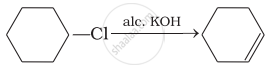
(iv)
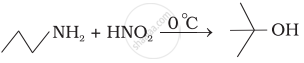
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.