Advertisements
Advertisements
प्रश्न
A radioactive nuclide `""_86^226` Ra decays by emission of two alpha particles, one beta particle and gamma rays. Which of the following is the resulting nuclide X?
पर्याय
223 x 83
218 x 83
218 x 84
217 x 85
218 x 85
उत्तर
Initially element is represented by 86Ra226
a particle is represented by 2He4 this means it contains 2 protons and 2
neutrons so mass of a particle is 4 unit. Thus after emitting a particle mass number of element would decrease by 4 unit and atomic number of element would decrease by 2.
So after emitting 2 a particle 86Ra226 would become 86 - 2 - 2Ra 226 - 4 - 4 = 82Ra218.
β particle is represented by -1e° after emitting a β particle atomic number of element would increase by 1 unit and mass number of element would remain unchanged.
So after emitting a p particle 82Ra218 would become 82 + 1Ra218 =83Ra218
Gamma radiations have zero mass and no charge so after emission of gamma radiation mass and atomic number remains unaltered.
The daughter nucleus is represented by b x a
b x a = 83Ra218
So answer is B.
APPEARS IN
संबंधित प्रश्न
Answer the following questions based on a hot cathode ray tube
Name the charged particles
In a cathode ray tube state:
(i) the purpose of covering cathode by thorium and carbon.
(ii) the purpose of the fluorescent screen.
(iii) how is it possible to increase the rate of emission of electrons.
In the following fig. shows a radioactive source S in a thick lead container. The radiations pass through an electric field between the plates P and Q. Complete the diagram to show the paths of α , β and γ radiations.
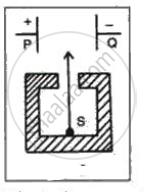
Why is the source S placed in a thick lead container?
Define the term radioactivity. Of the three normally occurring radioactive radiations:
(a) Which has a positive charge?
(b) Which is most penetrating?
(c) Which has no electric charge?
Name two radioactive substances.
A radioactive source emits three types of radiations.
Name the radiation which has the highest ionizing power.
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
Name a common device where a hot cathode ray tube is used.
A radioctive sample decays `7/8` times its original quantity in 15 minutes. The half-life of the sample is ______.
