Advertisements
Advertisements
प्रश्न
In a cathode ray tube state:
(i) the purpose of covering cathode by thorium and carbon.
(ii) the purpose of the fluorescent screen.
(iii) how is it possible to increase the rate of emission of electrons.
उत्तर
(i) The cathode is covered with thorium and carbon as it has low work function and it needs to be heated up to 2000 K to emit electrons. This serves it as a better electron emitter than tungsten.
(ii) The fluorescent screen helps in changing the electrical signal applied on the deflecting plates into the visual pattern on the screen.
(iii) The rate of emission of the electrons can be increased by
- Using a metal with a lower work function,
- Increasing the temperature of the surface of the metal, or
- Increasing the surface area of the metal emitting the electrons.
APPEARS IN
संबंधित प्रश्न
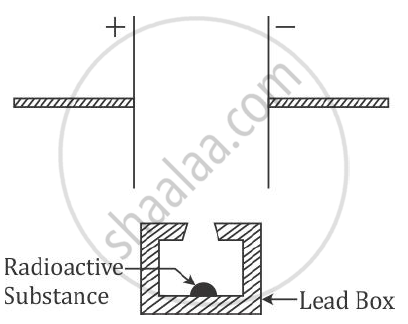
Complete the diagram as given above by drawing the deflection of radioactive radiations in an electric field
What are isobars?
What is meant by radioactivity?
A radioactive source emits three types of radiations. Name them.
(i) Name the radiations which are charged.
(ii) Name the radiations which are most penetrating.
(iii) Name the radiations which travel with the speed of light.
(iv) Name the radiations which have the largest mass.
Define bound electrons.
How is a cathode ray beam affected while passing through a magnetic field?
State any two uses of cathode rays.
In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
How is a cathode ray tube used to convert an electrical signal into a visual signal?
