Advertisements
Advertisements
प्रश्न
What is meant by radioactivity?
उत्तर
Elements with a high atomic number such as uranium, thorium, radium have a property of spontaneously emitting invisible, highly penetrating and high energy radiation. This property is called radioactivity. The nucleus of radioactive elements is unstable.
संबंधित प्रश्न
What are free electrons?
How can they be made to leave the metal surface? (State any two ways)
In a cathode ray tube, what is the function of anode?
Represent the change in the nucleus of a radioactive element when a β particle is emitted.
Why is a cathode ray tube evacuated to a low pressure?
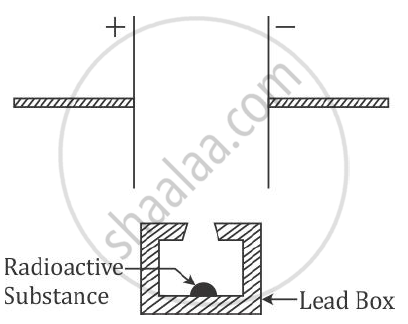
Complete the diagram as given above by drawing the deflection of radioactive radiations in an electric field
In a cathode ray tube state:
(i) the purpose of covering cathode by thorium and carbon.
(ii) the purpose of the fluorescent screen.
(iii) how is it possible to increase the rate of emission of electrons.
What do you mean by Mass number.
A radioactive source emits three types of radiations. Name them.
(i) Name the radiations which are charged.
(ii) Name the radiations which are most penetrating.
(iii) Name the radiations which travel with the speed of light.
(iv) Name the radiations which have the largest mass.
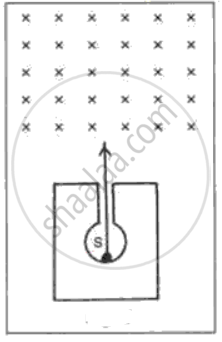
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
A certain nucleus A (mass number 238 and atomic number 92) is radioactive and becomes a nucleus B (mass number 234 and atomic number 90) by the loss of one particle.
What particle was emitted?
Why should a radioactive substance not be touched by hands?
Define atomic number of an element.
What do you mean by electron emission?
Name the three main parts of a Cathode Ray Tube.
How is a cathode ray tube used to convert an electrical signal into a visual signal?
A certain nucleus has a mass number 20 and atomic number 9. Find the number of neutrons and protons present in it.
An electrons emitter must have how much work function and melting point.
State three common properties of beta rays and cathode rays.
Mention four properties of Becquerel rays.
88Ra226 experiences three α - decay. Find the number of neutrons in the daughter element.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
A radioactive element is placed in an evacuated chamber. Then the rate of radioactive decay will ______.
Complete the following radioactive reaction:
\[\ce{^-_-X -> ^-_-Y + ^4_2He -> ^234_91Z + ^0_-1e}\]
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
