Advertisements
Advertisements
प्रश्न
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
उत्तर
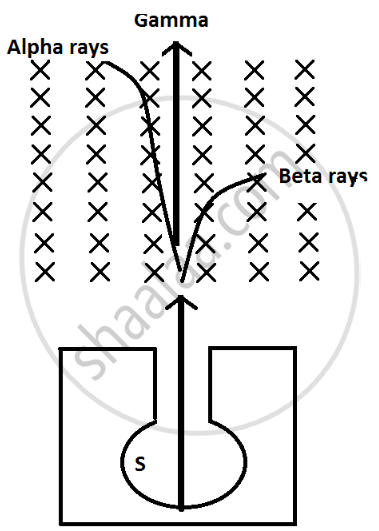
(i) Alpha particles are heavy in mass and are positively charged so they are deflected less by magnetic field and direction is upward which can be calculated by Fleming left hand rule.
(ii) Beta particles are negligible in mass so they are highly deflected by magnetic field and they are negatively charge particle so they are deflected in downward direction.
(iii) Gamma radiations have no mass and no charge so they are not deflected by magnetic field.
APPEARS IN
संबंधित प्रश्न
A radioactive substance is oxidized. Will there be any change in the nature its radioactivity? Give a reason for your answer
What are the uses of radioactive isotopes in following?
(a) Medical field,
(b) Agriculture,
(c) Industries?
A radioactive nucleus `""_"Z"^"A"` X first emits a beta particle and then an alpha particle to give the resulting nucleus `""_"Q"^"P"` Y What will be the values of P and Q in terms of A and Z?
A nucleus of an element X which has the symbol `""_84^202` X emits an alpha particle and then a beta particle. The final nucleus is `""_"b"^"a"` Y Find a and b.
Define free electrons.
How is a zinc sulphide screen affected when a cathode ray beam strikes it?
A radioactive nucleus \[\ce{^A_ZX}\] undergoes spontaneous decay in the sequence \[\ce{^A_ZX -> _{Z-1 }B -> _{Z-3}C -> _{Z-2}D }\], where Z is the atomic number of element X. The possible decay particles in the sequence are ______.
The half-life of a radioactive nuclide is 100 hours. The fraction of original activity that will remain after 150 hours would be ______.
Two radioactive materials A and B have decay constants 25λ and 16λ respectively. If initially they have the same number of nuclei, then the ratio of the number of nuclei of B to that of A will be "e" after a time `1/("a"lambda)` The value of a ______.
Complete the following radioactive reaction:
\[\ce{^-_-X -> ^-_-Y + ^4_2He -> ^234_91Z + ^0_-1e}\]
