Advertisements
Advertisements
Question
In following fig. shows a mixed source S of alpha and beta particles in a thick lead container. The particles pass through a magnetic field in a direction perpendicular to the plane of paper (inwards as shown by x). State and show in the diagram how the particles get affected.
Solution
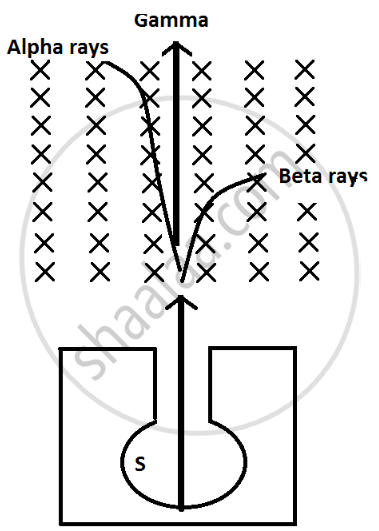
(i) Alpha particles are heavy in mass and are positively charged so they are deflected less by magnetic field and direction is upward which can be calculated by Fleming left hand rule.
(ii) Beta particles are negligible in mass so they are highly deflected by magnetic field and they are negatively charge particle so they are deflected in downward direction.
(iii) Gamma radiations have no mass and no charge so they are not deflected by magnetic field.
APPEARS IN
RELATED QUESTIONS
What do you mean by Mass number.
Name two radioactive substances.
During the emission of a beta particle, the ______ number remains same.
State two properties that a substance should possess when used as a thermionic emitter.
What are cathode rays? Why are they called ‘cathode rays’? Name a material which exhibits fluorescence when cathode rays fall on it.
In a cathode ray tube state. How is it possible to increase the rate of emission of electrons.
What do you understand by the term radioactivity?
A certain nucleus has a mass number 20 and atomic number 9. Find the number of neutrons and protons present in it.
In the following atoms, which one is a radioisotope? Give one use of this isotope.
O16, C14, N14, He4
