Advertisements
Advertisements
प्रश्न
A solid substance P which is very hard is used in the construction of many buildings, especially flooring. When substance P is heated strongly, it decomposes to form another solid Q and a gas R is given out. Solid Q reacts with water with the release of a lot of heat to form a substance S. When gas R is passed into a clear solution of substance S, then a white precipitate of substance T is formed. The substance T has the same chemical composition as starting substance P.
(a) What is substance P? Write its common name as well as chemical formula.
(b) What is substance Q?
(c) What is gas R?
(d) What is substance S? What is its clear solution known as?
(e) What is substance T? Name any two natural forms in which substance T occurs in nature.
उत्तर
(a) Substance P is calcium carbonate. Its common name is limestone and its chemical formula is CaCO3.
(b) Substance Q is calcium oxide (CaO).
(c) Gas R is carbon dioxide gas (CO2).
(d) Substance S is calcium hydroxide [Ca(OH)2]. Its clear solution is known as lime water.
(e) Substance T is calcium carbonate (CaCO3). Two natural forms of calcium carbonate are calcite and mollusc shell.
APPEARS IN
संबंधित प्रश्न
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
What type of chemical reaction take place when silver bromide is exposed to sunlight?
What type of chemical reaction is represented by the following equation?
X → Y + Z
Explain the following type of chemical reaction, giving two examples for it:
Decomposition reaction
Give scientific reason.
When the gas formed on heating limestone, is passed through freshly prepared lime water, the lime water turns milky.
Identify the type of following reaction :
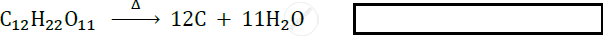
Give a balanced equation for –
A double decomposition neutralization reaction involving an acid and a base
Which one of the following processes involves chemical reactions?
Balance the following chemical equation and identify the type of chemical reaction.
`"H"_2"O"_2("l") overset("U V")(->) "H"_2"O"("l") + "O"_2("g")`
These consist of two statements – Assertion (A) and Reason (R). Answer these questions selecting the appropriate option given below:
Assertion: Silver bromide decomposition is used in black and white photography.
Reason: Light provides energy for this exothermic reaction.
