Advertisements
Advertisements
प्रश्न
A solution of caustic soda (NaOH) in water or when fused, conducts an electric current. what is the similarity in these two cases?
उत्तर
Caustic soda (NaOH) in water or in a fused state conducts electricity due to the dielectric effect of the polar water molecules, which overcomes electrostatic attraction and allows the ions to move freely.
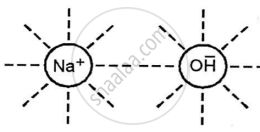
The high temperature needed to melt the solid weakens the bond between the particles in the molten state (fused), releasing the ions.
APPEARS IN
संबंधित प्रश्न
Differentiate between the terms strong electrolyte and weak electrolyte. (stating any two differences)
When fused lead bromide is electrolysed, we observe ______.
Substances which conduct electricity in the solid state are generally ______.
Name a non-metallic element which is a conductor of electricity.
Write the equations for the reactions, which takes place at the electrodes during the electrolysis of lead bromide?
If a fused metallic chloride is electrolyzed, at which electrode would the metal be obtained?
What kind of particles will be found in a liquid compound which is a non- electrolyte?
Here is an electrode reaction:
\[\ce{Cu -> Cu^{2+} + 2e^-}\]
At which electrode (anode or cathode) would such a reaction take place? Is this an example of oxidation?
Select the ion, that would get selectively discharge from the aqueous mixture of the ions listed below :
Pb2+, Ag+, Cu+
What is the difference between:
Strong electrolyte and weak electrolyte.
