Advertisements
Advertisements
प्रश्न
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
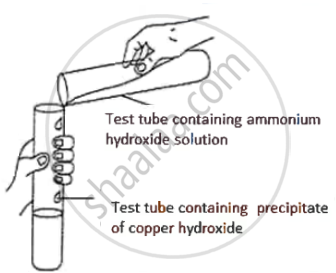
उत्तर
Blue ppt. dissolves to form an inky blue/deep blue solution.
APPEARS IN
संबंधित प्रश्न
Identify the substance underlined, in the following case
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide
Name the kind of particles present in sodium hydroxide solution.
What do you observe when caustic soda solution is added to the following solution, first a little and then in excess:
FeCl3
Write a balanced equation for this reaction.
Using sodium hydroxide solution, how will you distinguish: Zinc nitrate solution from calcium nitrate solution
Sodium hydroxide solution is added first in a small quantity, then in excess to the aqueous salt solutions of copper (II) sulphate, zinc nitrate, lead nitrate, calcium chloride, and iron (III) sulphate. Copy the following table and write the colour of the precipitate in (i) to (v) and the nature of the precipitate (soluble or insoluble) in (vi) to (x).
| Aqueous salt solution | Colour of precipitate when NaOH is added in a small quantity | Nature of precipitate (soluble or insoluble) when NaOH is added in excess |
| Copper (II) sulphate | (i) | (vi) |
| Zinc nitrate | (ii) | (vii) |
| Lead nitrate | (iii) | (viii) |
| Calcium chloride | (iv) | (ix) |
| Iron (III) Sulphate | (v) | (x) |
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to calcium salt solution and lead salt solution.
Distinguish by adding:
Sodium hydroxide solution and ammonium hydroxide solution to lead nitrate solution and zinc nitrate solution.
What is observed when hot, concentrated caustic soda solution is added to zinc? Write a balanced equation.
What is observed when hot, concentrated caustic soda solution is added to aluminium? Write a balanced equation.
Name the metal hydroxides which are insoluble in caustic soda solution.
