Advertisements
Advertisements
प्रश्न
A white powdery substance having strong smell of chlorine is used for disinfecting drinking water supply at waterworks. Identify the substance. Give its chemical name and write the chemical reaction for its preparation.
उत्तर
Bleaching powder is a white powder that emits a strong smell of chlorine and is used for disinfecting drinking water supply at waterworks.
The chemical name of bleaching powder is calcium oxychloride (CaOCl2).
The chemical reaction that takes place in its preparation is as follows.
Ca(OH)2 + Cl2 → CaOCl2 + H2O
APPEARS IN
संबंधित प्रश्न
Give the molecular formula of bleaching powder.
What is the common name of the compound CaOCl2?
Write the chemical name of bleaching powder and write its properties.
State two uses each of the following compounds:
Chlorine
Consider the following substance:
NaCl, Ca(OH)2, NaHCO3, NH3, Na2CO3, H2O, Cl2, CO2, CaSO4.2H2O, 2CaSO4.H2O, CaOCl2
Which two substance combine to form bleaching powder?
Give a scientific explanation.
Bleaching powder has the odour of chlorine.
Write the chemical formula.
Bleaching powder
Brine is an ____________.
The reaction between MnO2 with HCl is depicted in the following diagram. It was observed that gas with bleaching abilities was released.
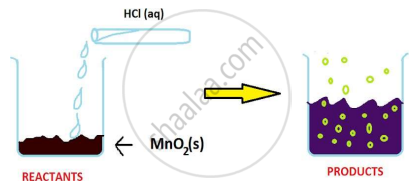
The chemical reaction between MnO2 and HCl is an example of:
Assertion (A): Reaction of Quicklime with water is an exothermic reaction.
Reason (R): Quicklime reacts vigorously with water releasing a large amount of heat.
