Advertisements
Advertisements
प्रश्न
Give a scientific explanation.
Bleaching powder has the odour of chlorine.
उत्तर
Chlorine is the main ingredient in bleaching powder. When exposed to air, bleaching powder reacts with carbon dioxide in the air to produce calcium carbonate and chlorine. Thus, it smells of chlorine.
CaOCl2 + CO2 `->` CaCO3 + Cl2
APPEARS IN
संबंधित प्रश्न
Give the molecular formula of bleaching powder.
What is the common name of the compound CaOCl2?
Write the chemical name of bleaching powder and write its properties.
Which compound of calcium is used for disinfecting drinking water supply?
Name one compound of calcium which is used for removing the colour of a coloured cloth.
Name the substance obtained by the action of chlorine on solid (dry) slaked lime.
State two uses each of the following compounds:
Chlorine
What is the common name of the compound CaOCl2?
How is bleaching powder prepared? Write chemical equation of the reaction involved in the preparation of bleaching powder.
What happens when bleaching powder reacts with dilute sulphuric acid? Give equation of the reaction involved.
State two important uses of bleaching powder.
Which of the following is treated with chlorine to obtain bleaching powder?
(a) CaSO4
(b) Ca(OH)2
(c) Mg(OH)2
(d) KOH
When chlorine and sodium hydroxide being produced during the electrolysis of brine are allowed to mix, a new chemical is formed. Name the chemical and write its uses.
Write the chemical formula.
Bleaching powder
Brine is an ____________.
CaOCl2 will liberate Cl2 gas in the presence of
(i) CO2
(ii) HCl
(iii) CO
(iv) NO
The reaction between MnO2 with HCl is depicted in the following diagram. It was observed that gas with bleaching abilities was released.
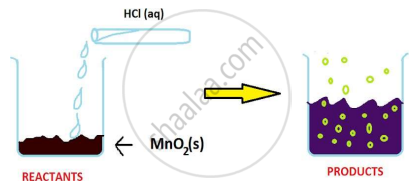
The chemical reaction between MnO2 and HCl is an example of:
Chlorine gas reacts with ____________ to form bleaching powder.
