Advertisements
Advertisements
प्रश्न
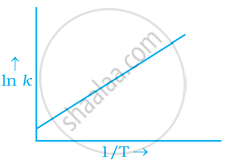
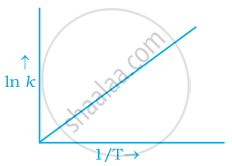
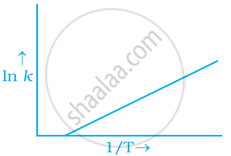
According to Arrhenius equation rate constant k is equal to `A e^((-Ea)/(RT)`. Which of the following options represents the graph of ln k vs `1/T`?
पर्याय
MCQ
उत्तर
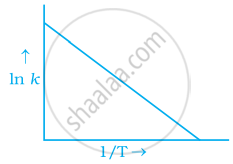
Explanation:
To arrhenius equation `k = A e^((-Ea)/(RT)`
Taking log on both sides in `k` = In `A.e^(-(E_a)/(RT))`
In `k` = In `A - (E_a)/(RT)`
In `k = - (-E_a)/R xx 1/T` + In A
y = mx + c
This equation can be related to equation of straight line.
From the graph, it is very clear that slope of the plot = `(-E_a)/R` and intercept = In A.
shaalaa.com
या प्रश्नात किंवा उत्तरात काही त्रुटी आहे का?