Advertisements
Advertisements
Question
According to Arrhenius equation rate constant k is equal to
Options
MCQ
Solution
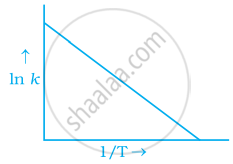
Explanation:
To arrhenius equation
Taking log on both sides in
In
In
y = mx + c
This equation can be related to equation of straight line.
From the graph, it is very clear that slope of the plot =
shaalaa.com
Is there an error in this question or solution?