Advertisements
Advertisements
Question
Consider a first order gas phase decomposition reaction given below :
\[\ce{A(g) -> B(g) + C(g)}\]
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as ______.
Options
`k = 2.303/t log p_i/(p_i - x)`
`k = 2.303/t log p_i/(2p_i - p_t)`
`k = 2.303/t log p_i/(2p_i + p_t)`
`k = 2.303/t log p_i/(p_i + x)`
Solution
The initial pressure of the system before decomposition of A was pi. After lapse of time ‘t’, total pressure of the system increased by x units and became ‘pt’ The rate constant k for the reaction is given as `k = 2.303/t log p_i/(2p_i - p_t)`.
Explanation:
Consider a first order gas phase decomposition reaction:
\[\ce{A_{(g)} -> B_{(g)} + C_{(g)}}\]
The initial pressure of the system before decomposition of A was Pi.
After lapse of time (t), total pressure of the system increased by x units and became 'Pt'.
In other words, the pressure of A decreased by x atom.
\[\ce{A_{(g)} -> B_{(g)} + C_{(g)}}\]
Initial pressure: Pi atm
Pressure after time t: (Pi – x) atm x atm x atm
Pt = (Pi – x) + x + x = Pi + x atm
x = Pt – Pi
PA = Pressure of A after time t = Pi - x = Pi - (Pi - Pi) = 2Pi - Pt
`k = 2.303/t log (["A"]_0)/(["A"])`
= `k = 2.303/t log p_i/(2p_i - p_t)`
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
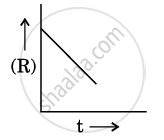
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
A reaction is first order in A and second order in B. Write the differential rate equation.
Molecularity of a reaction _____________.
The value of rate constant of a pseudo first order reaction ______.
Consider the reaction A ⇌ B. The concentration of both the reactants and the products varies exponentially with time. Which of the following figures correctly describes the change in concentration of reactants and products with time?
Assertion: Rate constants determined from Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason: Reactant molecules undergo chemical change irrespective of their orientation during collision.
Which of the following statement is true?
