Advertisements
Advertisements
प्रश्न
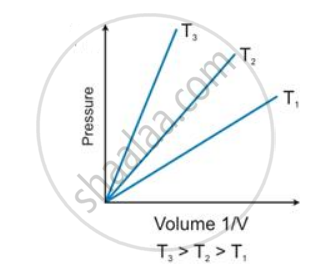
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is _______.
उत्तर
According to Boyle’s law, the shape of the graph between pressure and reciprocal of volume is Straight line.
APPEARS IN
संबंधित प्रश्न
State the law which is represented by the following graph:
A steel cylinder of internal volume 20 litres is filled with hydrogen at 29 atmospheric pressure. If hydrogen is used to fill a balloon at 1.25 atmospheric pressure at the same temperature, what volume will the gas occupy?
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
22.4 litres of gas weighs 70 g at STP. Calculate the weight of the gas if it occupies a volume of 20 litres at 27°C and 700 mmHg of pressure.
50 cm3 of hydrogen is collected over water at 17°C and 750 mmHg pressure. Calculate the volume of a dry gas at STP. The water vapour pressure at 17°C is 14 mmHg.
A gas cylinder having a capacity of 20 litres contains a gas at 100 atmos. How many flasks of 200 cm3 capacity can be filled from it at 1 atmos. pressure if the temperature remains constant?
Name or state the following:
The law which studies the relationship between pressure of a gas and the volume occupied by it at constant temperature.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1000 cc. originally occupying 1500 cc. at 720 mm. pressure.
